Over 1,000 Chemotherapy Protocols And Counting
New chemotherapy protocols are added weekly. All protocols are reviewed by Medical Oncologists and Hematologists.

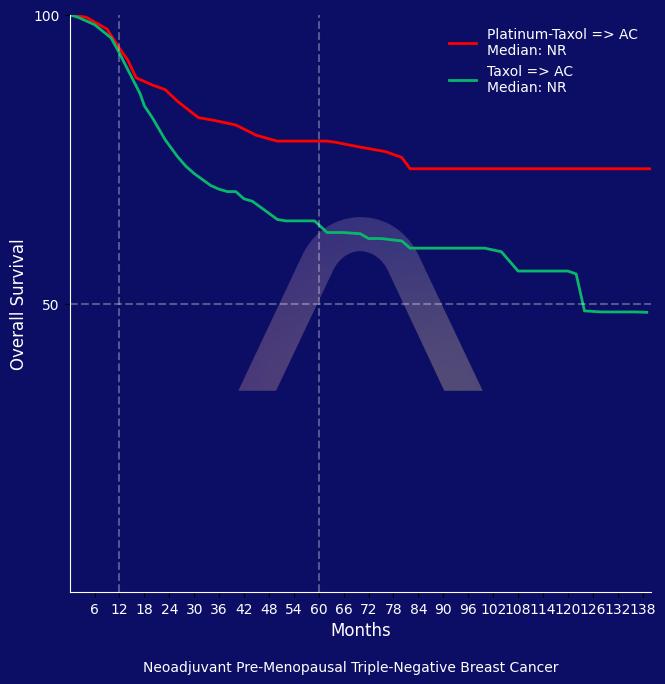
New Reference: Neoadjuvant Carboplatin for Triple-Negative Breast Cancer
This phase III trial evaluated the addition of carboplatin to neoadjuvant chemotherapy in triple-negative breast cancer. The addition improved overall survival, particularly in pre
New Reference: Nirogacestat for Desmoid Tumors
Long-term treatment with nirogacestat resulted in an objective response rate of 45.7%. Safety findings showed a decrease in the incidence and severity of treatment-emergent adverse
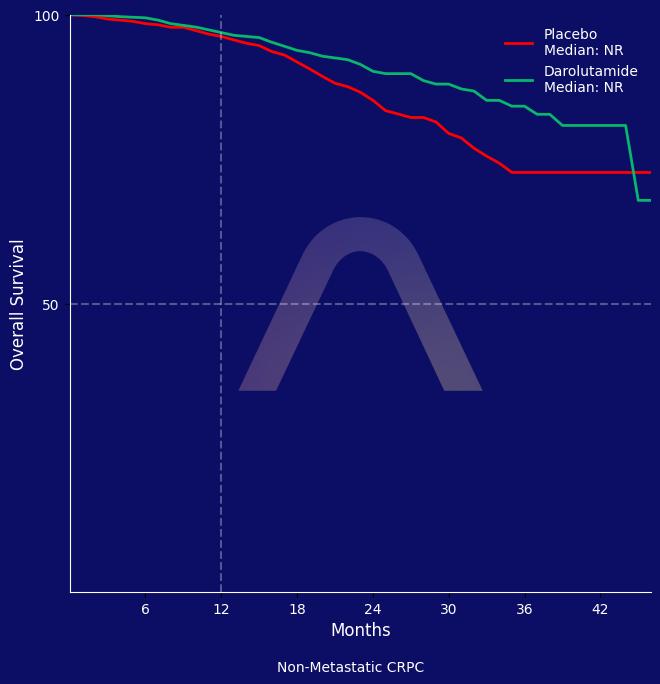
New Reference: Darolutamide for Non-Metastatic Castration Resistant Prostate Cancer
In this phase 3 trial, darolutamide significantly prolonged metastasis-free survival in men with nonmetastatic, castration-resistant prostate cancer compared to placebo, reducing t
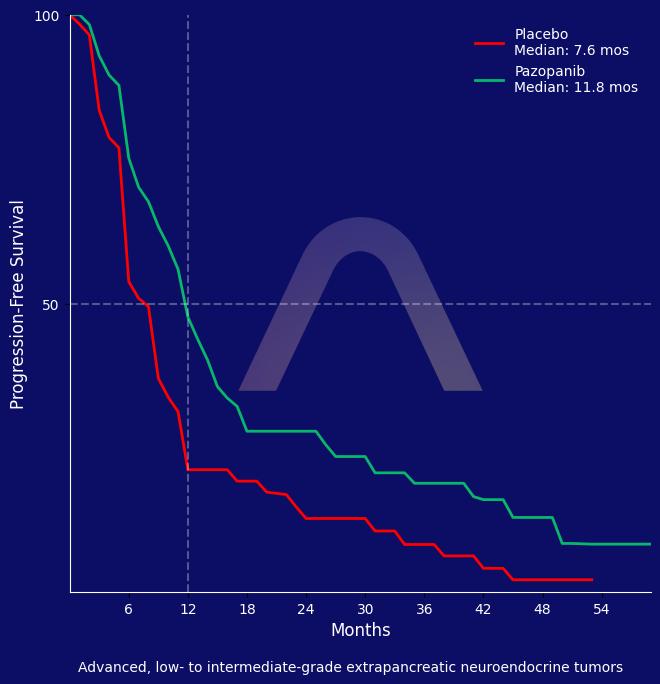
New Reference: Pazopanib for Extrapancreatic Neuroendocrine Tumors
Pazopanib demonstrated activity in prolonging progression-free survival for advanced extrapancreatic neuroendocrine tumors. However, it was associated with an increased rate of sev
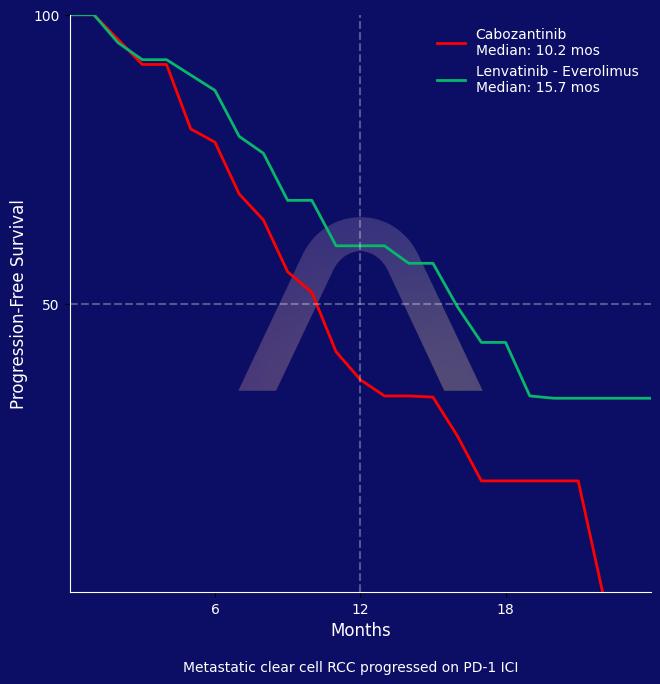
New Reference: Lenvatinib – Everolimus for Metastatic Clear Cell RCC
Lenvatinib plus everolimus prolonged progression-free survival compared to cabozantinib in patients with metastatic clear cell renal cell carcinoma that progressed on prior PD-1 in
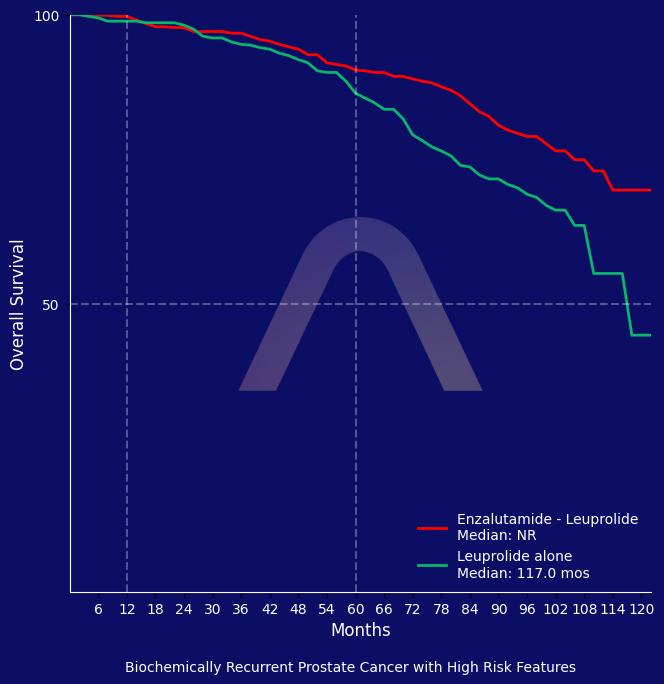
New Reference: Enzalutamide in Biochemically Recurrent Prostate Cancer
The trial showed that enzalutamide plus leuprolide significantly improved overall survival compared to leuprolide alone in patients with high-risk biochemically recurrent prostate
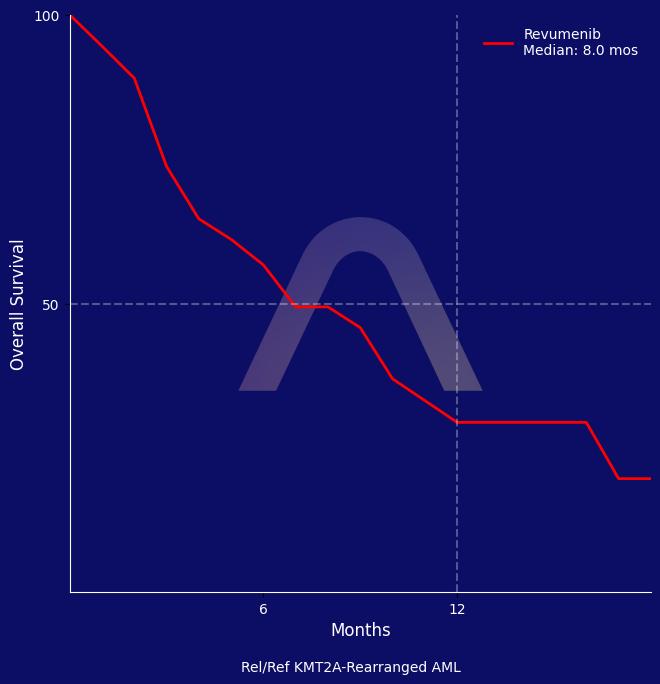
New Reference: Revumenib for Rel/Ref KMT2A-Rearranged AML
The AUGMENT-101 study showed that revumenib led to high response rates in relapsed or refractory KMT2A-rearranged acute leukemia. Patients had an overall response rate of 63.2% wit
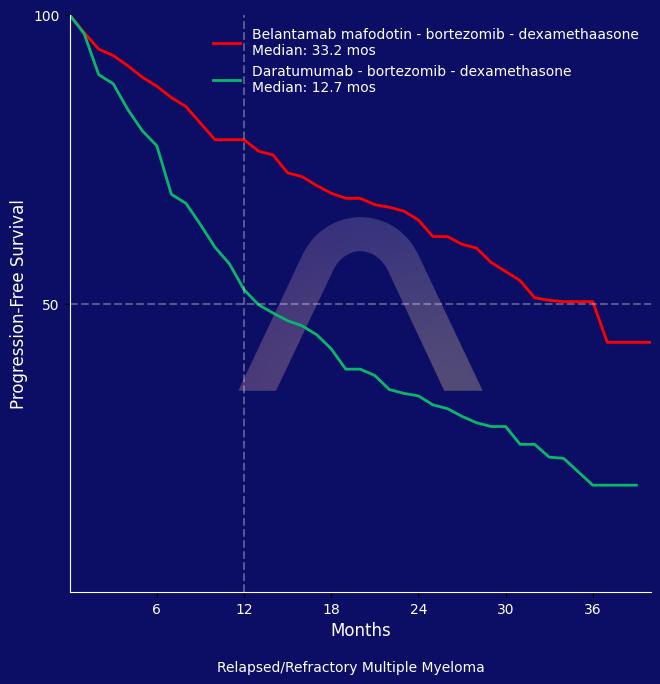
New Reference: Belantamab for Multiple Myeloma
BVd therapy significantly prolonged progression-free survival compared to DVd in patients with relapsed or refractory multiple myeloma. It was associated with a higher rate of comp
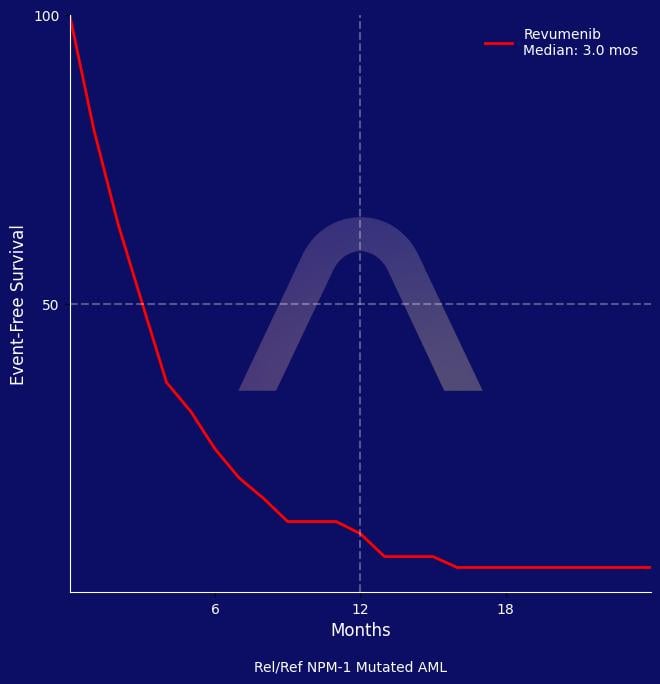
New Indication: Revumenib for Rel/Ref NPM1-Mutated AML
In this study, revumenib exhibited a promising overall response rate of 46.9% in patients with relapsed or refractory NPM1-mutated acute myeloid leukemia. The median overall surviv
Easiest Access to Our Chemotherapy Protocol Library
Download our mobile app on iOS or Android to easily access our cancer protocol library, calculate chemotherapy doses, staging library, and access to the reference summaries