Over 1,000 Chemotherapy Protocols And Counting
New chemotherapy protocols are added weekly. All protocols are reviewed by Medical Oncologists and Hematologists.

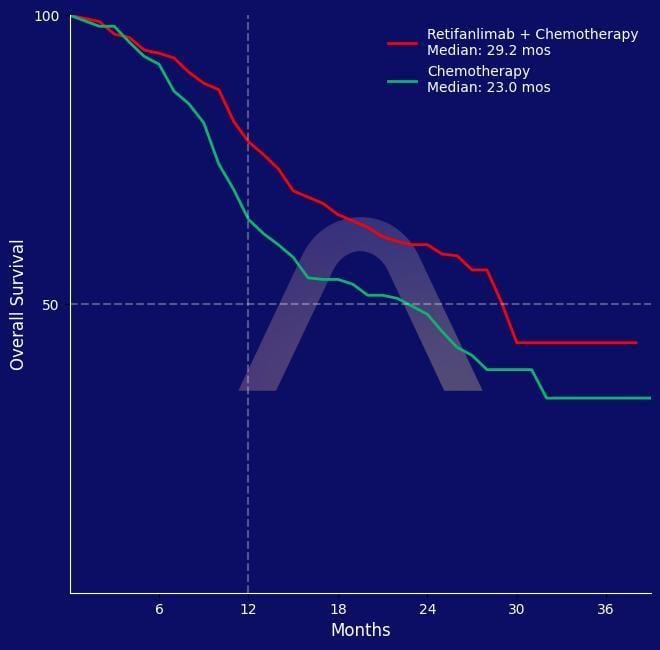
New Reference: Retifanlimab + Chemotherapy for Advanced Anal Cancer
Randomised, double-blind, phase 3 trial [POD1UM-303/InterAACT-2]. Patients with inoperable locally recurrent or metastatic squamous cell carcinoma of the anal canal, aged >=18, ECO
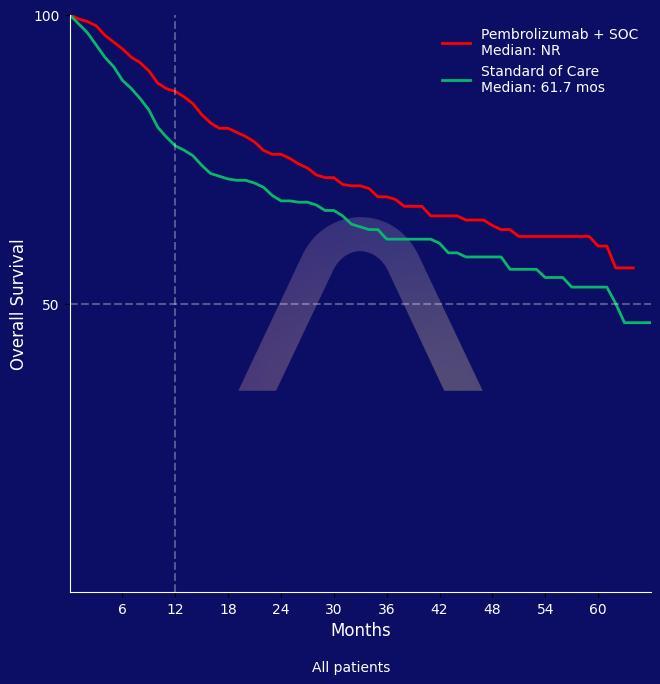
New Indication: Periop Pembrolizumab for Head and Neck Cancer
Phase 3, open-label, randomized, multicenter trial [KEYNOTE-689] Locally advanced resectable head and neck squamous-cell carcinoma (HNSCC) Neoadjuvant and adjuvant pembrolizumab pl
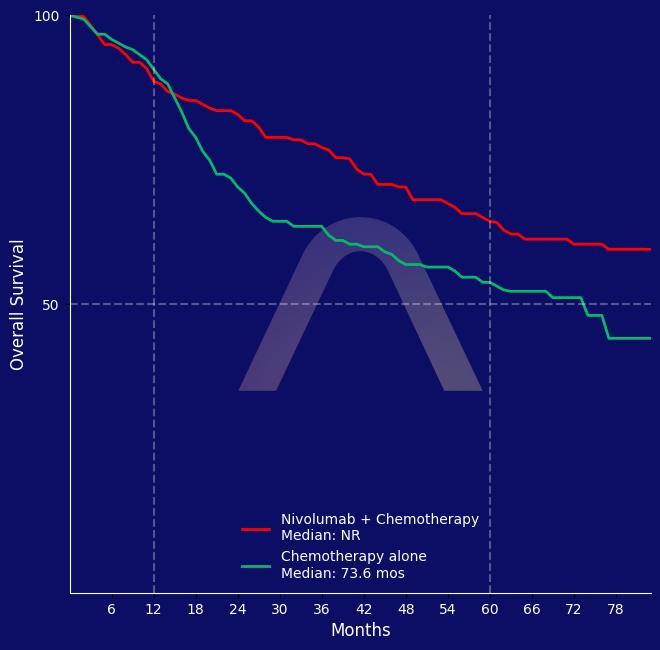
New Reference: Neoadjuvant Nivolumab for NSCLC
Randomized, open-label, phase 3 trial [CheckMate 816] Resectable stage IB to IIIA NSCLC Nivolumab+chemotherapy (n=179) vs chemotherapy (n=179) for 3 cycles followed by surgery
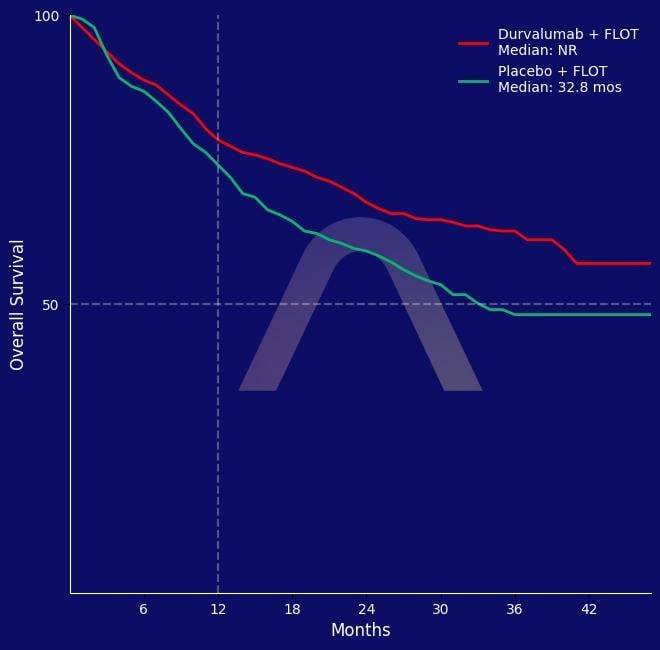
New Protocol: Perioperative Durvalumab with FLOT in Gastric Cancer
Phase 3, multinational, double-blind, randomized trial [MATTERHORN] Resectable gastric or gastroesophageal junction adenocarcinoma Durvalumab (n=474) vs Placebo (n=474) every 4 wee
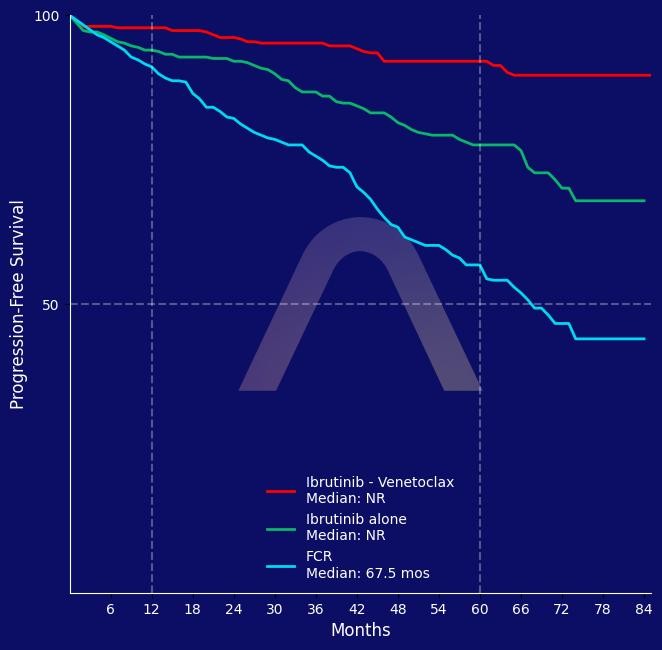
New Reference: MRD-Guided CLL Treatment
Phase 3, multicenter, open-label trial [FLAIR ISRCTN01844152] Previously untreated chronic lymphocytic leukemia (CLL) patients Ibrutinib-venetoclax vs. Ibrutinib alone vs. Fludarab
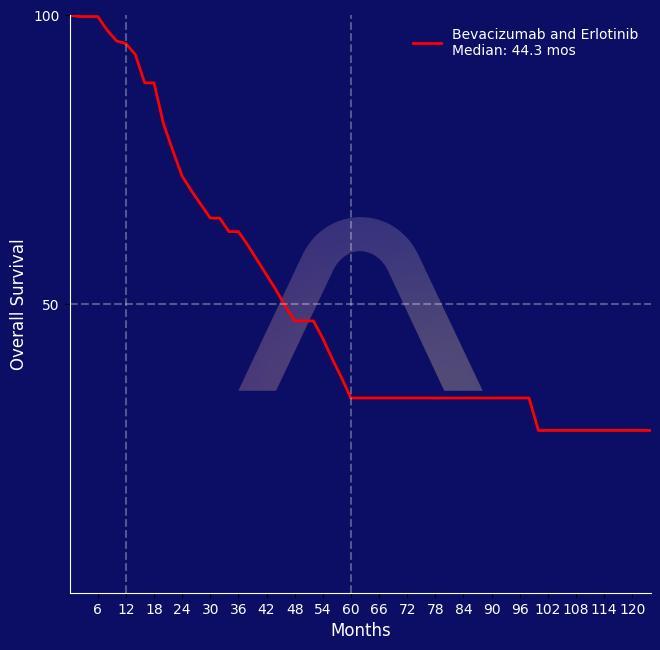
New Indication: Bevacizumab and Erlotinib for Papillary Renal Cell Carcinoma
Open-label, phase 2 study [NCT01130519] Advanced HLRCC-associated or sporadic papillary renal-cell carcinoma Bevacizumab (10 mg/kg every 2 wks) + Erlotinib (150 mg daily)
New Reference: Maintenance Atezolizumab for Stage IB-IIIA NSCLC
Open-label, randomized, phase 3 trial (IMpower010) Completely resected stage IB-IIIA NSCLC after adjuvant platinum-based chemotherapy Atezolizumab (1,200 mg q3w for 16 cycles) vs.
New Protocol: Maintenance Sacituzumab Govitecan with Avelumab for Urothelial Carcinoma
Phase 2, randomized, open-label trial (JAVELIN Bladder Medley) Locally advanced or metastatic urothelial carcinoma without progression after first-line platinum-based chemotherapy
New Indication: Daratumumab for High-Risk Smoldering Myeloma
Phase 3, open-label, multicenter, randomized trial (AQUILA) Patients with high-risk smoldering multiple myeloma without end-organ damage Daratumumab subcutaneous monotherapy vs. ac
Easiest Access to Our Chemotherapy Protocol Library
Download our mobile app on iOS or Android to easily access our cancer protocol library, calculate chemotherapy doses, staging library, and access to the reference summaries