Over 1,000 Chemotherapy Protocols And Counting
New chemotherapy protocols are added weekly. All protocols are reviewed by Medical Oncologists and Hematologists.

New Reference: Advanced Pancreatic Cancer Protocols
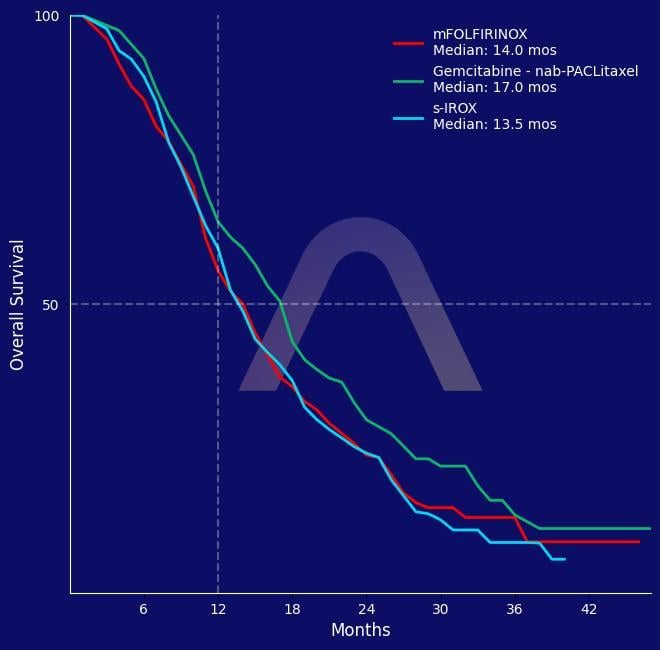
Randomized, open-label, phase II/III trial [GENERATE, JCOG1611] Metastatic or recurrent pancreatic cancer mFOLFIRINOX (n=175) vs S-IROX (n=176) vs nab-paclitaxel gemcitabine (n=176
New Protocol: Brentuximab – ECADD Chemo for Hodgkin Lymphoma
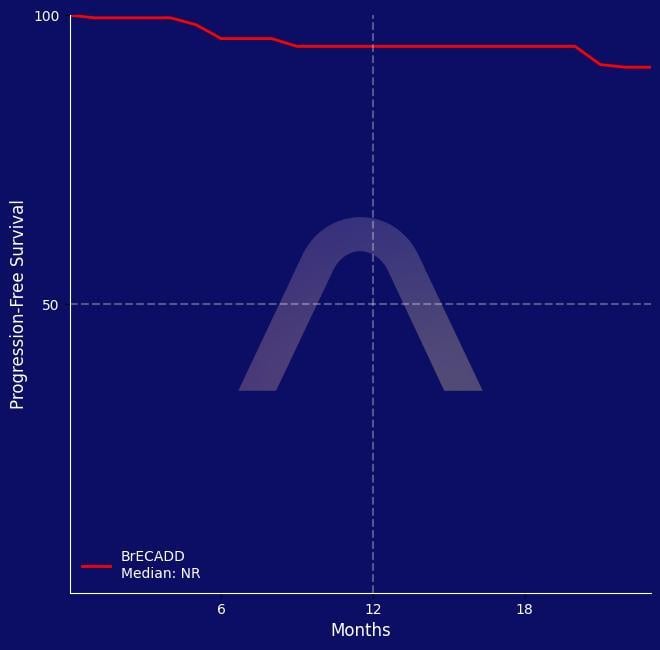
Prospective, multicenter, single-arm, phase II cohort study [HD21 trial, NCT02661503] Older patients with advanced-stage classic Hodgkin lymphoma aged 61-75 years 4-6 cycles of PET
New Indication: Neoadjuvant Osimertinib with/without Chemo for NSCLC
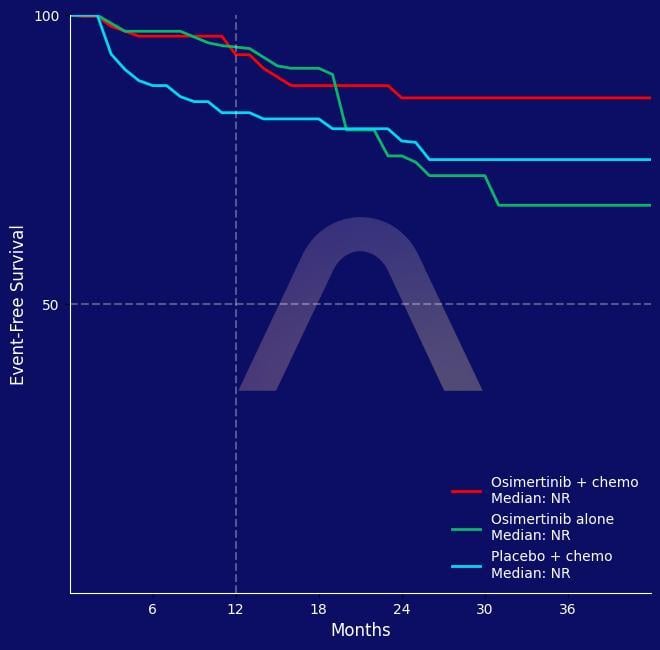
Randomized, controlled, phase III study [NeoADAURA] NCT04351555 Resectable, EGFR-mutated, stage II-IIIB NSCLC Osimertinib+chemotherapy (n=121) vs Osimertinib (n=117) vs Placebo+che
New Protocol: Glofitamab with R-CHOP for High-Risk DLBCL
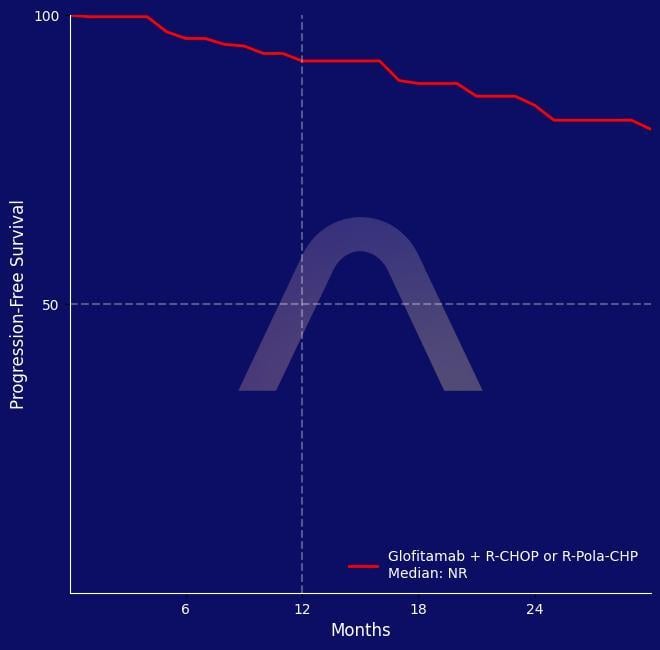
Phase II, open-label, multicenter study [COALITION; NCT04914741] Younger patients (≤65 years) with high-risk large B-cell lymphoma R-CHOP (n=40) vs Pola-R-CHP (n=40) with Glofi
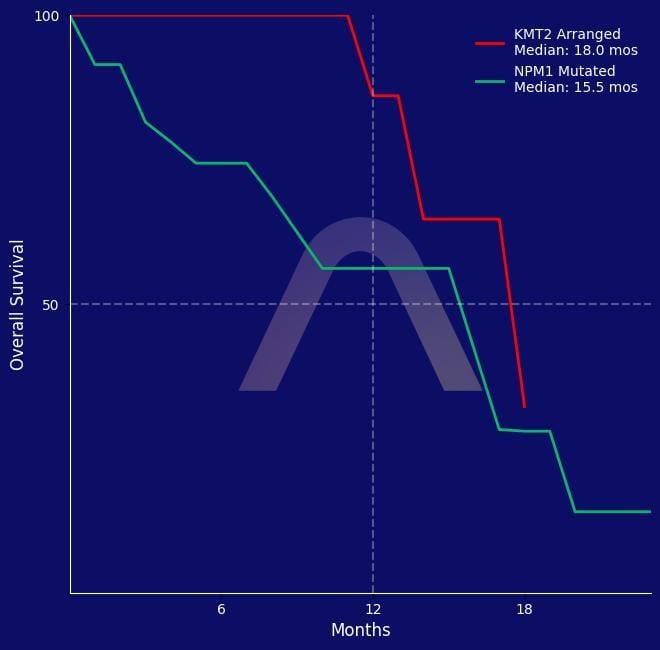
New Protocol: Revumenib with Azacitidine and Venetoclax for KMT2-Rearranged / NPM1-Mutated
Phase I dose-escalation and expansion study [NCT03013998] First-line treatment for patients aged 60 years and older with newly diagnosed NPM1-mutated or KMT2A-rearranged AML Azac
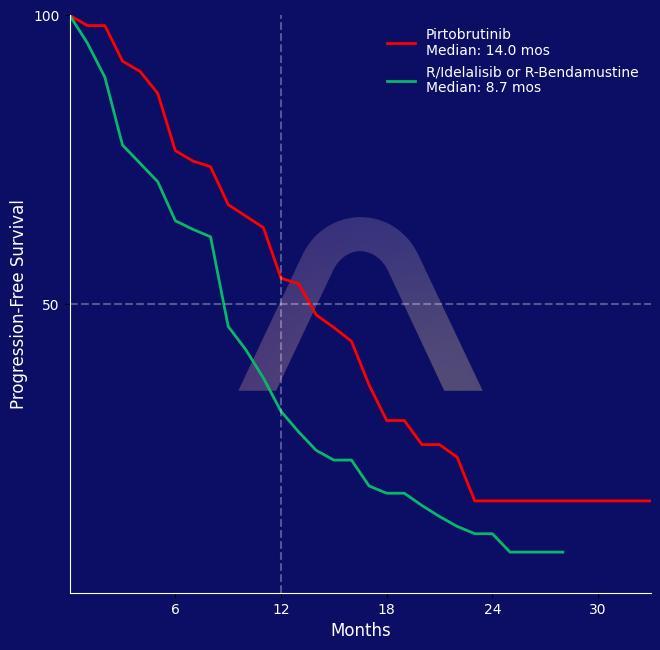
New Reference: Pirtobrutinib for BTKi Pretreated CLL
Open-label, randomized, phase III study [BRUIN CLL-321] Relapsed/refractory CLL/SLL, previously treated with BTKi Pirtobrutinib (n=119) vs investigator's choice of Idelalisib/Ritux
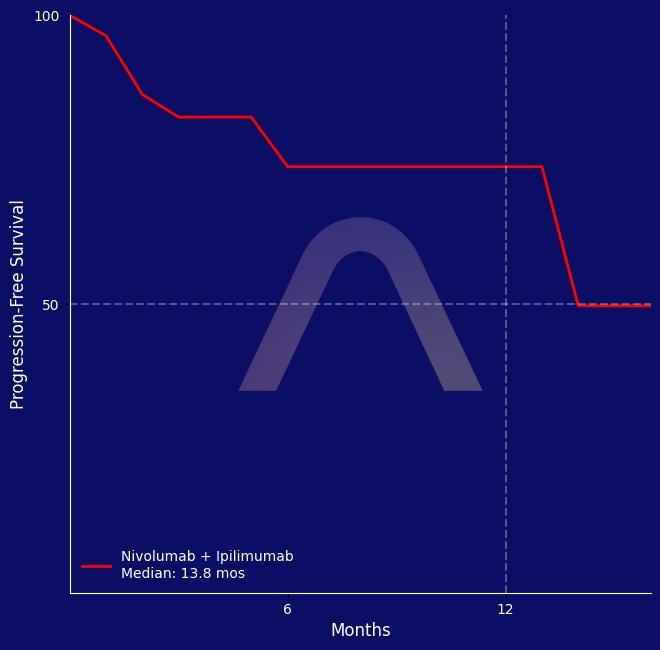
New Reference: Nivolumab and Ipilimumab for Advanced Gastric Cancer
Single-arm phase II study [NO LIMIT, WJOG13320G]. Microsatellite instability-high advanced gastric or esophagogastric junction cancer. Nivolumab (240 mg every 2 weeks) plus low-dos
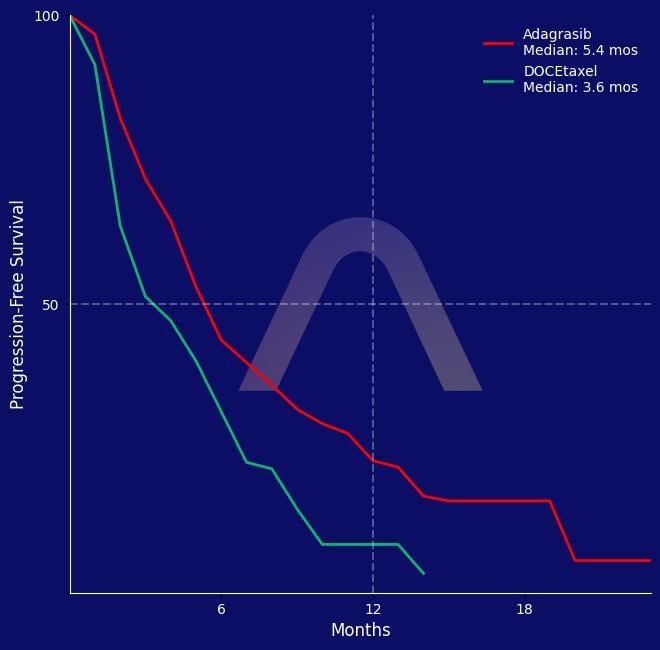
New Reference: Adagrasib for kRAS Mutated NSCLC
Randomised, open-label, phase 3 trial [KRYSTAL-12] Advanced NSCLC with KRASG12C mutation, previously treated with chemotherapy and immunotherapy 600 mg adagrasib twice daily (n=301
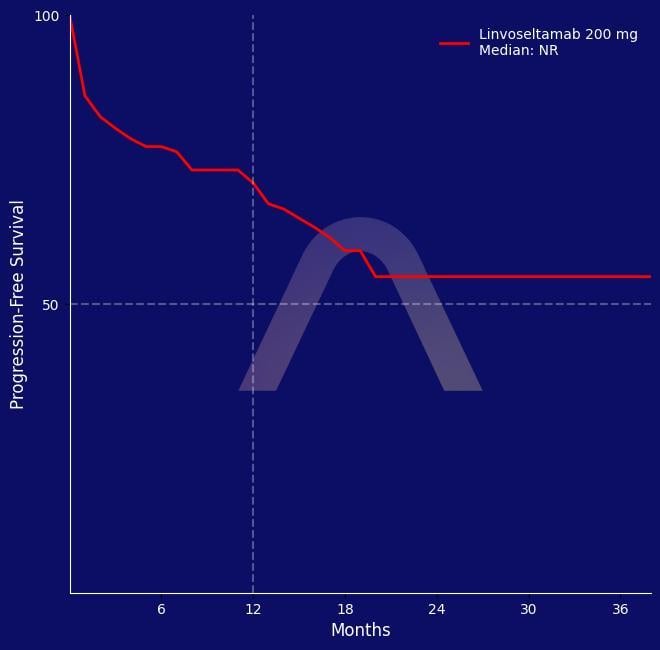
New Drug: Linvoseltamab for Relapsed/Refractory Myeloma
Phase I/II, first-in-human trial Relapsed/refractory multiple myeloma Livoseltamab 50mg (n=104) vs 200mg (n=117)
Easiest Access to Our Chemotherapy Protocol Library
Download our mobile app on iOS or Android to easily access our cancer protocol library, calculate chemotherapy doses, staging library, and access to the reference summaries