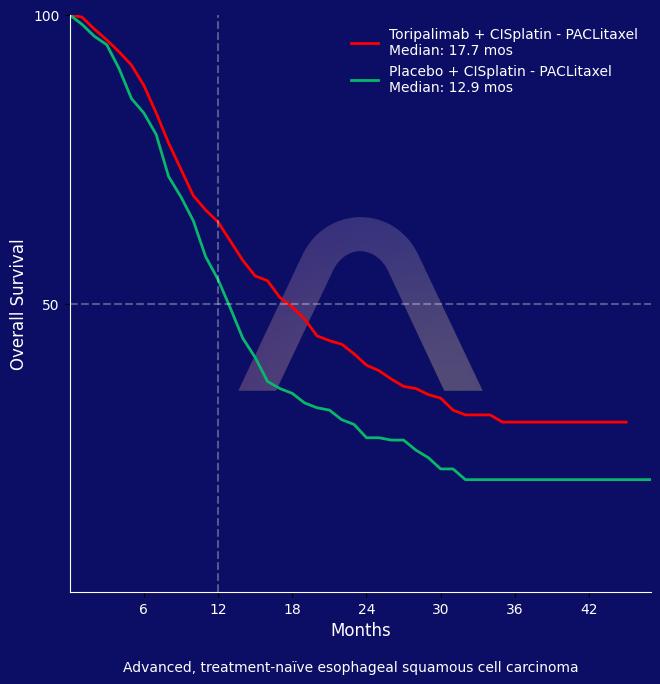
In the JUPITER-06 trial, toripalimab combined with chemotherapy significantly improved overall and progression-free survival in patients with advanced esophageal squamous cell carcinoma. The treatment was well tolerated, with similar safety profiles between the groups.
Study
|
Randomized, double-blind, placebo-controlled, phase 3 study [JUPITER-06] |
| Advanced, treatment-naïve esophageal squamous cell carcinoma not eligible for curative surgery or definitive chemoradiation |
| Toripalimab plus CISplatin + PACLitaxel (n=257) vs Placebo plus CISplatin + PACLitaxel (n=257) every 3 weeks for up to 6 cycles followed by maintenance toripalimab vs. placebo
|
Efficacy
|
mPFS: 7.0 mos vs 5.6 mos (HR 0.61 [0.49-0.74]) |
| mOS: 17.7 mos vs 12.9 mos (HR 0.72 [0.58-0.88]) |
| 3-yr OS: 29.7% vs 19.9%
|
Safety
|
Grade >=3 AEs: 75.1% vs 71.2% |
| Leukopenia (68.1% vs 53.7%), Neutropenia (67.7% vs 55.6%), Thrombocytopenia (30.0% vs 17.1%), Rash (24.9% vs 10.5%) |
| Deaths due to AEs: 9.3% vs 9.3%
|
Ann Oncol. Published online 2026
http://doi.org/10.1016/j.annonc.2026.01.005
Reviewed by Ulas D. Bayraktar, MD on Feb 19, 2026