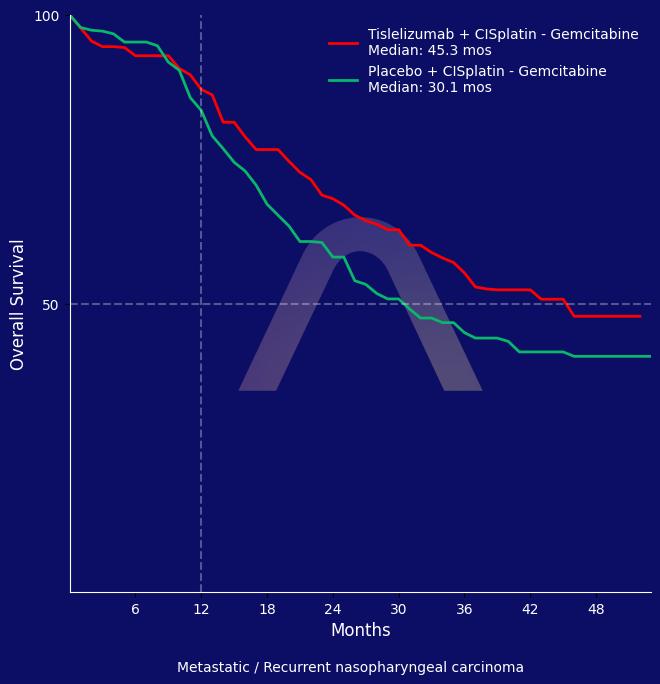
The RATIONALE-309 trial demonstrated that tislelizumab plus chemotherapy provided sustained progression-free survival and numerical overall survival improvements compared to placebo plus chemotherapy in recurrent or metastatic nasopharyngeal carcinoma. While progression-free survival improvement was modest, the overall survival benefit was significant. Safety profile was consistent with known immune-related risks.
Study
|
Double-blind, placebo-controlled phase 3 randomized clinical trial [RATIONALE-309] |
| Recurrent or metastatic nasopharyngeal carcinoma, treatment-naive |
| Tislelizumab+chemotherapy (CISplatin – gemcitabine) (n=131) vs placebo+chemotherapy (n=132)
|
Efficacy
|
mPFS: 9.6 mos vs 7.4 mos (HR 0.53 [0.39-0.71]) |
| mOS: 45.3 mos vs 31.8 mos (HR 0.73 [0.51-1.05])
|
Safety
|
Grade >=3 AEs: 85.0% vs 85.4% |
| Serious AEs: 35.3% vs 35.4% |
| Treatment discontinuations: 16.5% vs 10.8% |
| Immune-mediated AEs (any grade): 53.4% vs 37.7%
|
JAMA Oncol. Published online February 26, 2026
http://doi.org/10.1001/jamaoncol.2026.0020
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026