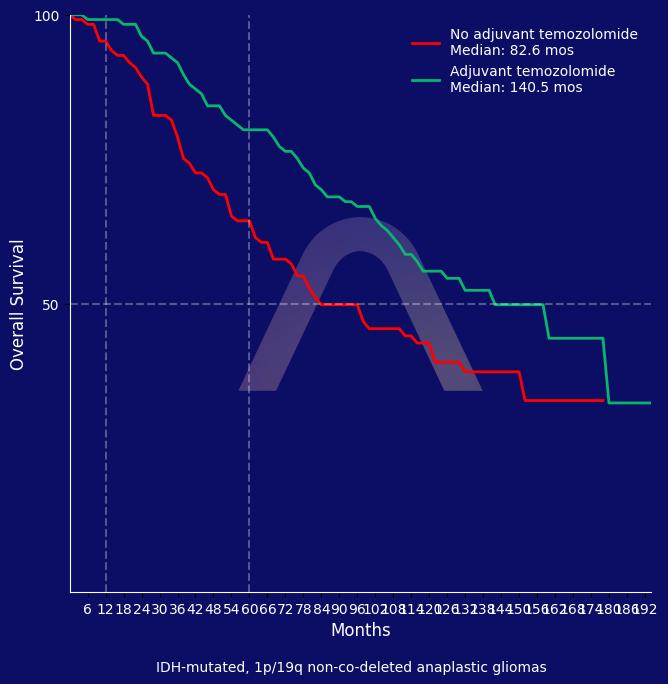
The CATNON trial demonstrated a survival benefit with adjuvant temozolomide in patients with IDH-mutated astrocytoma compared to radiotherapy alone. Concurrent temozolomide did not show any additional overall survival benefit. This long-term analysis supports the use of 12 cycles of adjuvant temozolomide for IDH-mutated 1p/19q non-co-deleted gliomas
Study
|
Randomised, open-label, phase 3 trial [CATNON; EORTC 26053-22054] |
| Adult patients with newly diagnosed 1p/19q non-co-deleted anaplastic gliomas (WHO 200 classification) |
| Radiotherapy (RT) alone vs RT with concurrent temozolomide vs. RT with adjuvant temozolomide vs. RT with both concurrent/adjuvant temozolomide
|
Efficacy
|
Cox model showed improved OS with adjuvant temozolomide but not with concurrent temozolomide (HR: 0.65 [0.54-0.77]) |
| In subset analyses, only among IDH mutated pts adjuvant temozolomide improved OS (HR: 0.54 [0.42-0.69])
|
Safety
|
Not reported
|
Lancet Oncol 2026;27:45-56
http://doi.org/10.1016/S1470-2045(25)00544-X
Reviewed by Ulas D. Bayraktar, MD on Feb 19, 2026