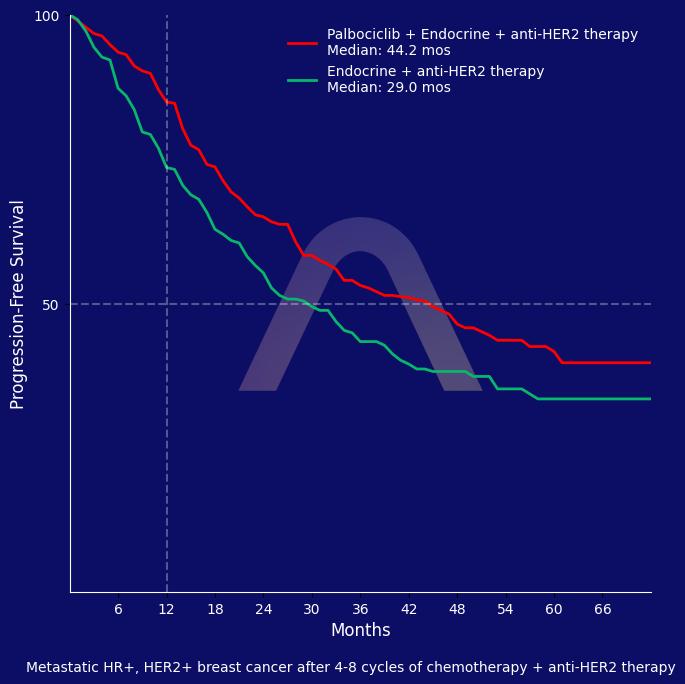
The addition of palbociclib to maintenance anti-HER2 and endocrine therapies significantly improved progression-free survival in patients with hormone-receptor-positive, HER2-positive advanced breast cancer. However, this was accompanied by increased toxic effects, primarily neutropenia.
Study
|
Phase 3, open-label, randomized trial [PATINA] |
| Hormone-receptor–positive, HER2-positive metastatic breast cancer pts who did not have disease progression after 4-8 cycles of chemotherapy + her2-targeted therapy |
| Maintenance HER2-targeted and endocrine therapies with palbociclib (n=261) vs without palbociclib (n=257)
|
Efficacy
|
Confirmed response (pts who achieved CR with induction chemo excluded): 32.9% vs. 24.8% (with palbociclib vs. without palbociclib) |
| mPFS: 44.3 mos vs 29.1 mos (HR 0.75 [0.59-0.96])
|
Safety
|
Grade >=3 AE: Neutropenia (60.5% vs 2.0%), leukopenia (16.1% vs 0.8%), diarrhea (9.6% vs 1.2%) |
| Serious AEs: 28.7% vs 21.8%
|
N Engl J Med 2026;394:451-62
http://doi.org/10.1056/NEJMoa2511218
Reviewed by Ulas D. Bayraktar, MD on Feb 19, 2026