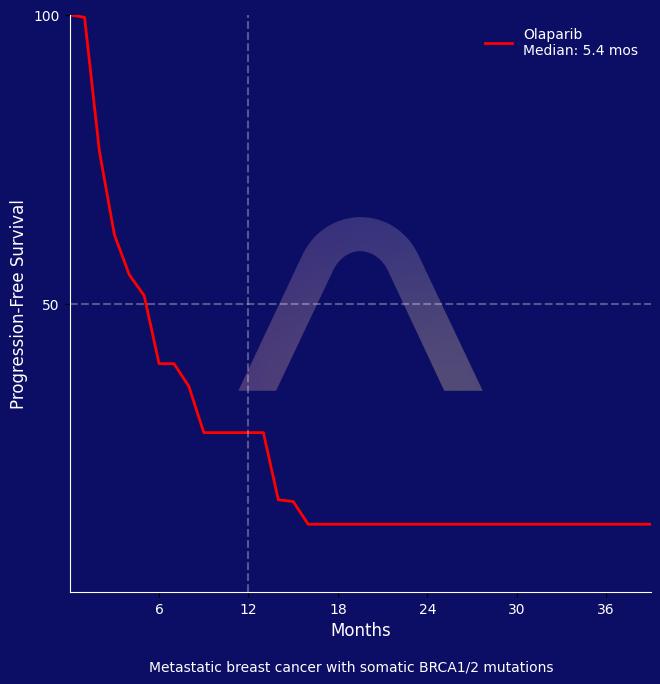
The phase II TBCRC 048 study found olaparib to be effective in metastatic breast cancer patients with germline PALB2 or somatic BRCA1/2 mutations. The overall response rate was significantly higher in those with PALB2 mutations compared to those with somatic BRCA mutations. Olaparib was well tolerated, with manageable side effects.
Study
|
Phase II study [TBCRC 048] |
| Metastatic breast cancer patients with germline PALB2 mutations or somatic BRCA1/2 mutations |
| Olaparib 300 mg twice daily until progression
|
Efficacy
|
ORR: 75% vs 36.7% (gPALB2m vs. sBRCAm) |
| Median PFS: 9.4 mos vs 5.5 mos |
| Median DOR: 7.0 mos vs 11.2 mos
|
Safety
|
Grade 3 anemia (13%), grade 2 alopecia (2%) |
| Dose reductions required in 40.7%; 1 discontinuation due to grade 3 anemia
|
J Clin Oncol 2026;00:1-9
http://doi.org/10.1200/JCO-25-02075
Reviewed by Ulas D. Bayraktar, MD on Feb 15, 2026