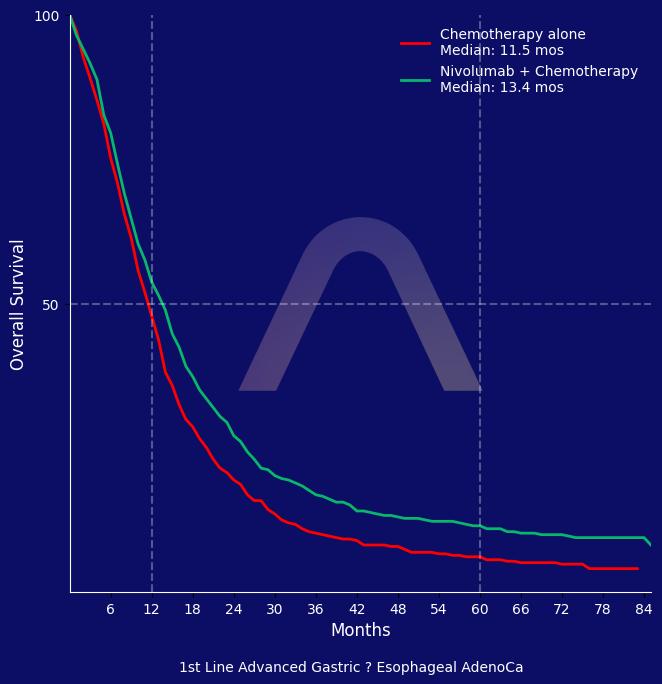
Nivolumab combined with chemotherapy continues to show superior overall survival and progression-free survival compared to chemotherapy alone at 5 years in advanced gastroesophageal adenocarcinoma. The treatment showed a higher objective response rate, and the safety profile was consistent with no new safety concerns reported.
Study
|
Randomized, open-label, phase 3 study [CheckMate 649] |
| Previously untreated, non-HER2+, unresectable or metastatic gastric, GE-junction, esophageal adenocarcinoma |
| Nivolumab+chemotherapy (n=789) vs chemotherapy (n=792)
|
Efficacy
|
ORR: 58% vs 46% (nivolumab vs. placebo) |
| mOS in all pts: 13.7 mos vs 11.6 mos (HR 0.79 [0.71-0.88]) |
| mPFS in all pts: 7.8 mos vs 6.9 mos (HR 0.79 [0.71-0.89]) |
| mOS in PD-L1 CPS >10%: 15 mos vs. 10.9 mos (HR 0.68 [0.58-0.79])
|
Safety
|
Grade >=3 AE: 60% vs 45% |
| Treatment discontinuation: 42% vs 26%
|
Ann Oncol. Published online 2026 Feb 6
http://doi.org/10.1016/j.annonc.2026.02.003
Reviewed by Ulas D. Bayraktar, MD on Feb 15, 2026