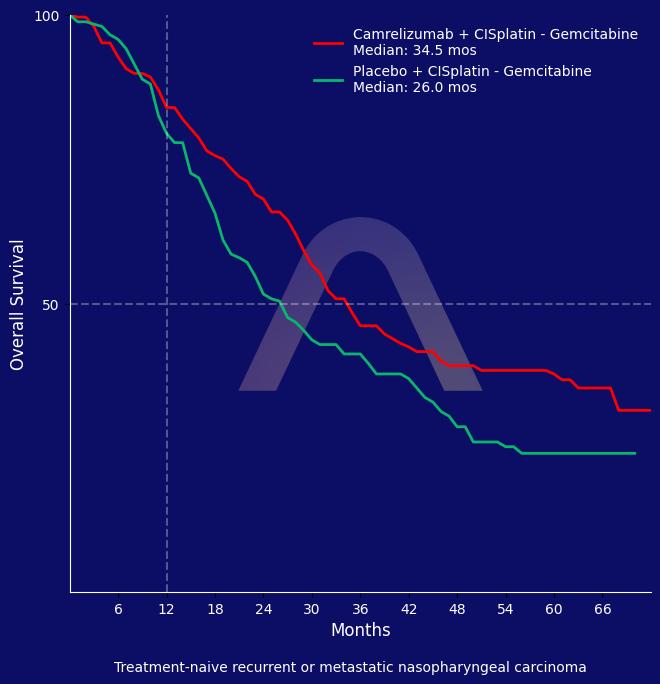
The addition of camrelizumab to chemotherapy provided significant improvement in overall survival in patients with recurrent or metastatic nasopharyngeal carcinoma. The trial demonstrated a median overall survival of 34.5 months compared to 26.6 months with chemotherapy alone, establishing camrelizumab as a standard of care in this setting.
Efficacy
|
mOS: 34.5 mos vs 26.6 mos (HR 0.74 [0.55-0.99]) |
| 5-yr OS: 37.8% vs 24.2%
|
Safety
|
Grade >=3 AEs: Not reported |
| Treatment discontinuation due to AEs: 20 patients (15%) in camrelizumab group vs 14 patients (11%) in placebo group. |
| Deaths due to AEs: 4 patients (3%) in camrelizumab group vs 3 patients (2%) in placebo group.
|
JAMA Oncol. Published online January 29, 2026
http://doi.org/10.1001/jamaoncol.2025.6245
Reviewed by Ulas D. Bayraktar, MD on Feb 15, 2026