The study found that adjuvant nivolumab significantly improved disease-free survival compared to placebo in patients with high-risk muscle-invasive urothelial carcinoma. The analysis also identified circulating tumor DNA as a potential biomarker for recurrence risk, with undetectable ctDNA levels indicating a better prognosis.
Study
|
Phase III randomized, double-blind, multicenter study [CheckMate 274] |
| Patients with high-risk muscle-invasive urothelial carcinoma after radical surgery |
| Nivolumab (n=353) vs placebo (n=356), every 2 weeks for up to 1 year
|
Efficacy
|
Median DFS: 21.9 mos vs 11.0 mos (nivolumab vs. placebo) (HR 0.74 [0.61-0.90]) |
| Median OS: 75.0 mos vs 50.1 mos (HR 0.83 [0.67-1.02]) |
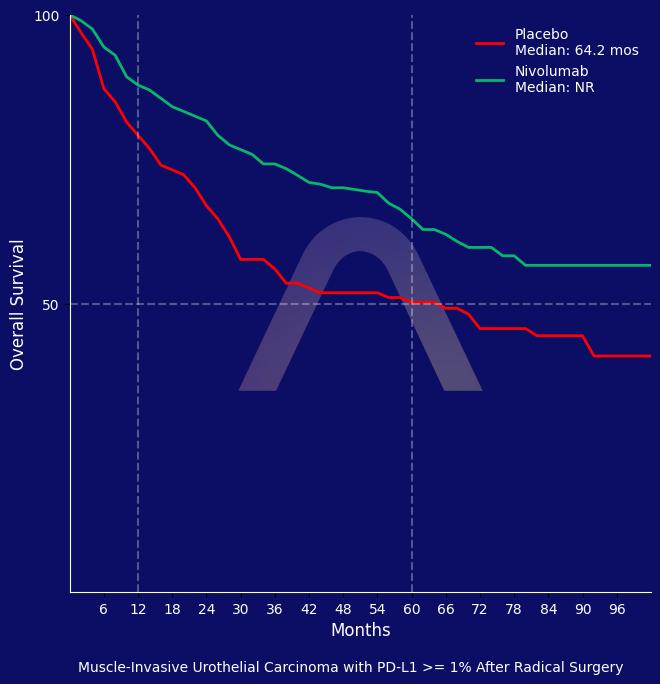
| Patients with PD-L1 >=1%: |
| Median DFS 55.5 mos vs 8.4 mos (HR 0.58 [0.42-0.79]) |
| Median OS: NR vs. 59.4 mos (HR: 0.63 [0.44-0.90])
|
Safety
|
No new safety signals reported. |
| Common treatment-related AEs included pruritus, fatigue, and diarrhea.
|
J Clin Oncol. Published online 17 October 2025
http://doi.org/10.1016/j.annonc.2025.09.139
Reviewed by Ulas D. Bayraktar, MD on Jan 21, 2026