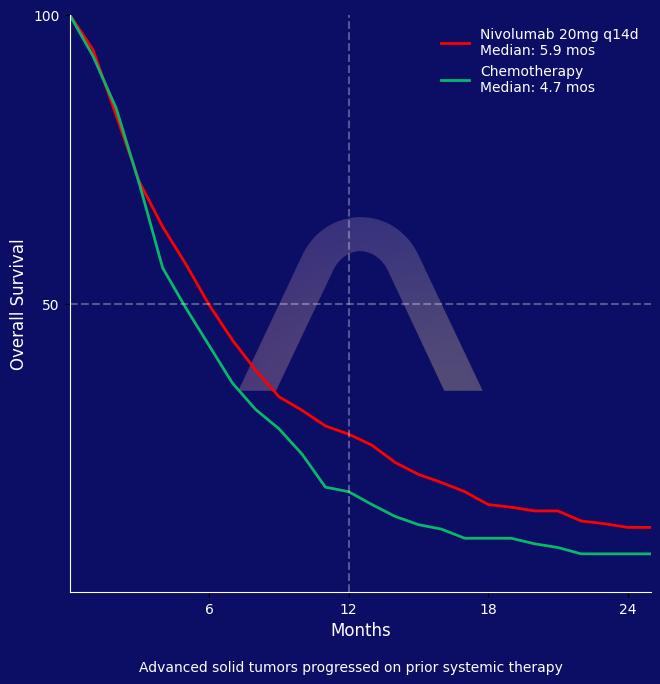
Ultra-low-dose nivolumab significantly improved overall survival compared to chemotherapy in patients with relapsed refractory solid tumors while reducing severe toxicities. The regimen did not improve progression-free survival or objective response rate but showed a better quality of life.Of importance, over 80% of enrolled patients had head/neck or lung cancer.
Study
|
Phase III randomized superiority trial [DELII] |
| Advanced solid tumors with progression on prior systemic therapy |
| Nivolumab 20 mg q2wks (n=250) vs standard chemotherapy (n=250) |
| Overall 51.8% of pts had H-N and 36.4% had lung cancer
|
Efficacy
|
ORR: 7.1% vs 8.1% |
| DCR: 37.7% vs 39.3% |
| mPFS: 2.04 mos vs 2.09 mos (HR 1.03 [0.86-1.23]) |
| mOS: 5.88 mos vs 4.70 mos (HR 0.80 [0.66-0.97]) |
| 1-yr OS: 27.3% vs 16.9%
|
Safety
|
Grade >=3 AE: anemia (21.5% vs 7.8%), neutropenia (7.6% vs 0.4%), diarrhea (10.5% vs 1.2%), hyponatremia (40.5% vs 28.6%), infection (11.0% vs 2.9%)
|
J Clin Oncol. Published online January 28, 2026
http://doi.org/10.1200/JCO-25-01546
Reviewed by Ulas D. Bayraktar, MD on Feb 15, 2026