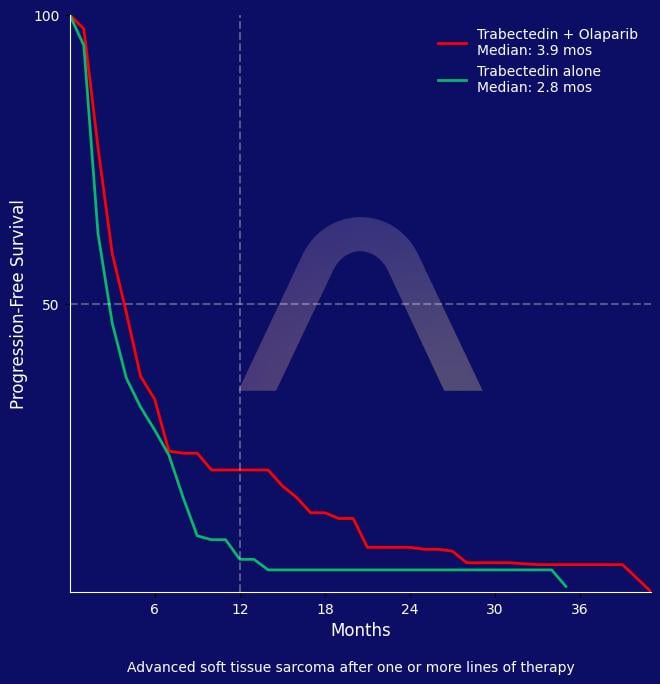
This phase II randomized study in advanced soft tissue sarcoma patients found a marginal progression-free survival benefit with trabectedin plus olaparib compared to trabectedin alone. Notably, patients with PARP1 positive tumors and those with uterine leiomyosarcoma derived substantial benefit from the combination. Hematological toxicities were more frequent with the combination but manageable, supporting further histology- and biomarker-driven investigation.
Study
|
Investigator-initiated, open-label, multicenter, phase II randomized clinical trial [TOMAS2; NCT03838744] |
| Advanced/metastatic soft tissue sarcomas progressing after one or more prior lines of therapy |
| Trabectedin – olaparib (n=65) vs trabectedin alone (n=62)
|
Efficacy
|
ORR: 12.7% vs 7.9% (combo vs. trabectedin alone) |
| 6mo-PFS: 32% [22-46] vs 28% [19-42] |
| mPFS: 3.9 mos [2.7-5.2] vs 2.9 mos [2.2-3.6] (HR 0.722 [0.501-1.041]) |
| In uterine leiomyosarcoma subgroup, 1yr-PFS: 42.9% vs 0% (HR 0.501 [0.203-1.236]) |
| In PARP1 positive patients, PFS6m: 41.5% vs 27.8%, mPFS: 4.3 mos vs 2.5 mos (HR 0.537 [0.337-0.855])
|
Safety
|
Grade >=3 adverse events: neutropenia (trabectedin-olaparib 35.4% vs trabectedin 38.5%) |
| Permanent treatment discontinuation due to AEs: 7.7% vs 4.6% |
| Dose reductions/delays more frequent with combination
|
Ann Oncol. 2026;37(4):521
http://doi.org/10.1016/j.annonc.2025.11.019
Reviewed by Ulas D. Bayraktar, MD on Apr 10, 2026