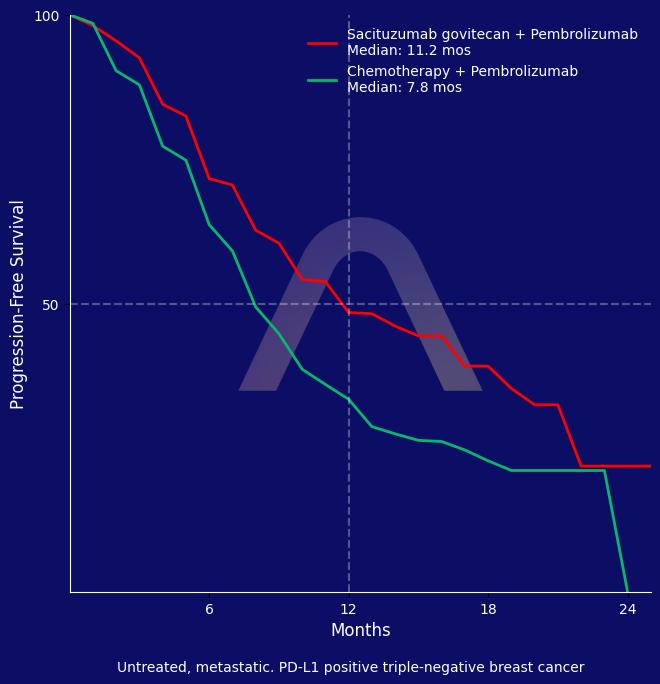
ASCENT-04 phase 3 trial showed that sacituzumab govitecan plus pembrolizumab significantly improved progression-free survival compared to chemotherapy plus pembrolizumab in patients with PD-L1-positive advanced triple-negative breast cancer. Objective response rates were higher, and responses were more durable with the combination treatment. Safety profiles were consistent with known effects of the drugs.
Study
|
Phase 3, open-label, international trial [ASCENT-04/KEYNOTE-D19] |
| Untreated, unresectable/metastatic triple-negative breast cancer, PD-L1-positive |
| Sacituzumab govitecan plus pembrolizumab (n=221) vs chemotherapy plus pembrolizumab (n=222)
|
Efficacy
|
ORR: 60% vs 53% (sacituzumab + pembro vs. chemo + pembro) |
| mPFS: 11.2 mos vs 7.8 mos (HR 0.65 [0.51-0.84]) |
| mDOR: 16.5 mos vs 9.2 mos |
| Overall survival immature
|
Safety
|
Grade >=3 AE: neutropenia (43% vs 45%), diarrhea (10% vs 2%), anemia (7% vs 16%) |
| Treatment discontinuation: 12% vs 31% |
| Deaths due to AEs: 3% vs 3%
|
Engl J Med 2026;394:354-66
http://doi.org/10.1056/NEJMoa2508959
Reviewed by Ulas D. Bayraktar, MD on Feb 19, 2026