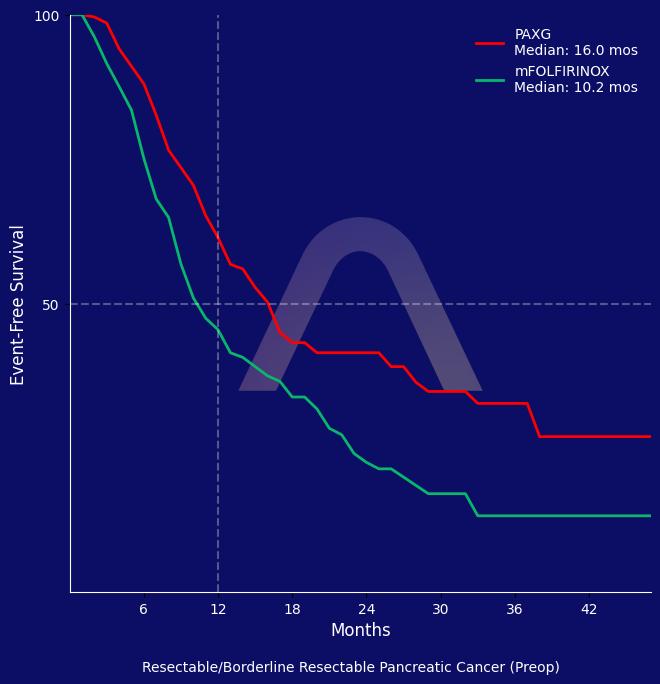
Preoperative PAXG significantly improved event-free survival compared to mFOLFIRINOX in resectable or borderline resectable pancreatic ductal adenocarcinoma. PAXG demonstrated a favorable safety profile with manageable toxicity and could be considered a new standard option for neoadjuvant therapy in this setting.
Study
|
Randomised, open-label, 2 × 2 factorial phase 3 trial [PACT-21 CASSANDRA] |
| Resectable and borderline resectable pancreatic ductal adenocarcinoma |
| PAXG (n=132) vs mFOLFIRINOX (n=128)
|
Efficacy
|
ORR: 46% vs. 39% (PAXG vs. mFOLFIRINOX) |
| 1-year EFS rate: 61% vs 45% |
| 3-year EFS rate: 33% vs 13% |
| mOS: 32.1 mos vs 26.4 mos
|
Safety
|
Grade >=3 AEs: 66% vs 61% |
| Neutropenia: 36% vs 24% |
| Treatment-related death: 0% vs 1%
|
Lancet. Published online November 20, 2025
Reni M, Macchini M, Orsi G New Protocol: Preoperative PAXG for Pancreatic Cancer
http://doi.org/10.1016/S0140-6736(25)01685-X
Reviewed by Ulas D. Bayraktar, MD on Dec 8, 2025