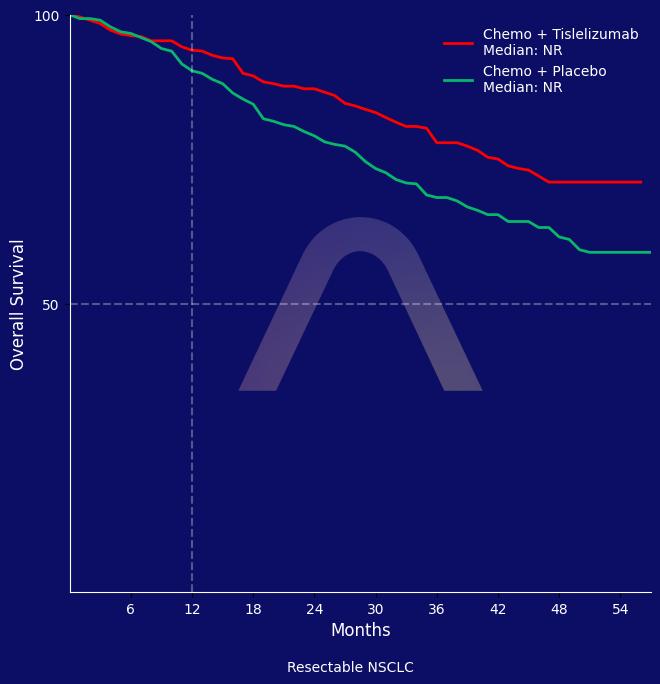
The RATIONALE-315 trial demonstrated that peri-operative tislelizumab with chemotherapy significantly improved overall survival and event-free survival compared to chemotherapy alone in patients with resectable non-small cell lung cancer. The treatment was generally well tolerated, though it showed a higher incidence of serious treatment-related adverse events.
Study
|
Randomized, double-blind, phase III trial [RATIONALE-315] |
| Resectable stage II-IIIA NSCLC |
| Tislelizumab+chemotherapy (n=226) vs placebo+chemotherapy (n=227)
|
Efficacy
|
36-month OS: 79.3% vs 69.3% (HR 0.65 [0.45-0.93]) |
| mEFS: NR vs 30.6 mos (HR 0.58 [0.43-0.79])
|
Safety
|
Grade >=3 TRAEs: 73.0% vs 67.3% |
| Serious TRAEs: 15.5% vs 8.8% |
| Deaths due to TRAEs: 1.8% vs 0.9%
|
Ann Oncol. Published online 2025
Wang C, Wang W, Liu H New Protocol: Periop Tislelizumab with Chemotherapy for Resectable NSCLC
http://doi.org/10.1016/j.annonc.2025.11.017
Reviewed by Ulas D. Bayraktar, MD on Dec 8, 2025