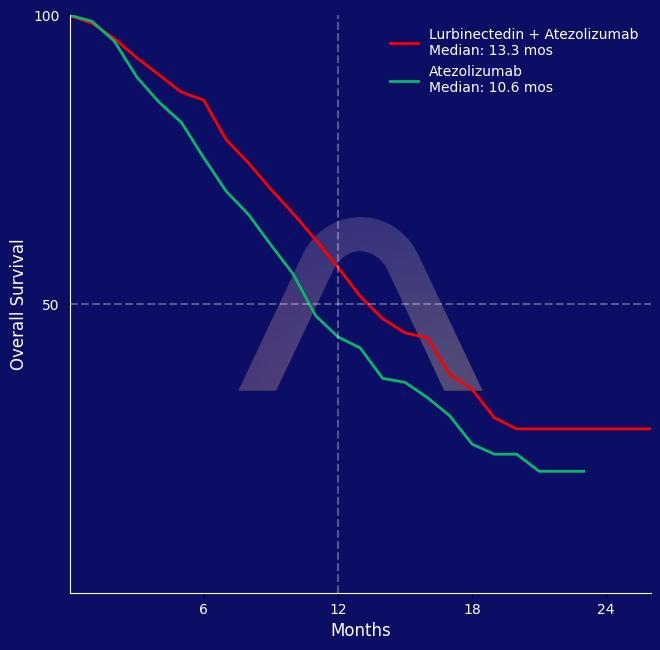
Study
| Randomized, multicentre, open-label, phase 3 trial [IMforte] |
| First-line maintenance therapy in extensive-stage small-cell lung cancer (ES-SCLC) |
| Lurbinectedin plus atezolizumab (n=242) vs atezolizumab (n=241) every 3 weeks |
Efficacy
| Progression-free survival: 5.4 mos vs 2.1 mos (HR 0.54 [0.43-0.67]) |
| Overall survival: 13.2 mos vs 10.6 mos (HR 0.73 [0.57-0.95]) |
Safety
| Grade >=3 AEs: anaemia (8% vs 1%), decreased neutrophil count (7% vs 0%), decreased platelet count (7% vs 0%) |
| Serious AEs: 31% vs 17% |
| Treatment discontinuation: 6% vs 3% |
Lancet Oncol. Published online June 2, 2025
http://doi.org/10.1016/S0140-6736(25)01011-6
Reviewed by Elvin Chalabiyev, MD on Sep 3, 2025