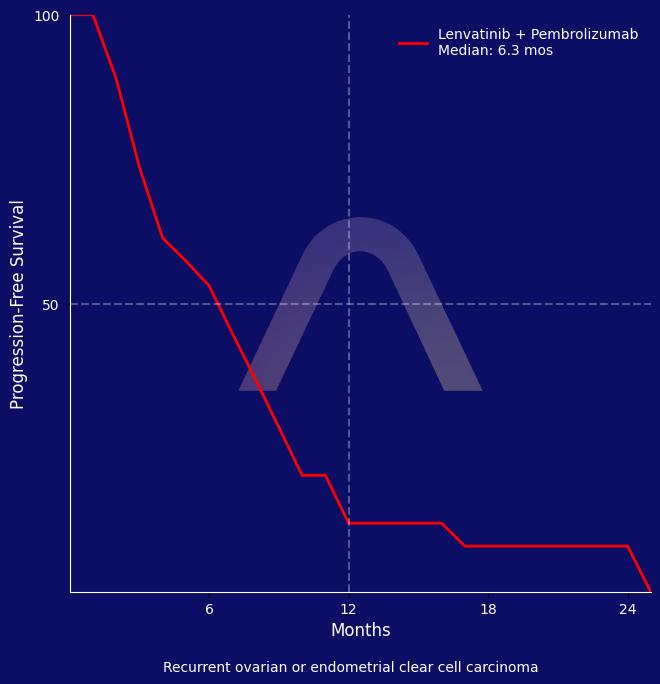
In this phase 2 trial of pembrolizumab plus lenvatinib in patients with recurrent gynaecological clear cell carcinoma, the combination of lenvatinib and pembrolizumab showed a 40% objective response rate and manageable safety
Study
|
Multicentre, single-arm, phase 2 trial [NCT04699071] |
| Recurrent ovarian or endometrial clear cell carcinoma, progressed or recurred after platinum-based chemotherapy |
| Pembrolizumab + Lenvatinib (n=27)
|
Efficacy
|
ORR: 40% [21-61] |
| mPFS: 6.4 mos [3.4-9.5] |
| mOS: 15.6 mos [7.9-not available]
|
Safety
|
Grade >=3 AEs: Hypertension (22%), decreased platelet count (7%), elevated AST (7%), immune-related hepatitis (7%) |
| Discontinuation due to toxicity: 7%
|
Lancet Oncol. Published online January 15, 2026
http://doi.org/10.1016/S1470-2045(25)00662-X
Reviewed by Ulas D. Bayraktar, MD on Feb 19, 2026