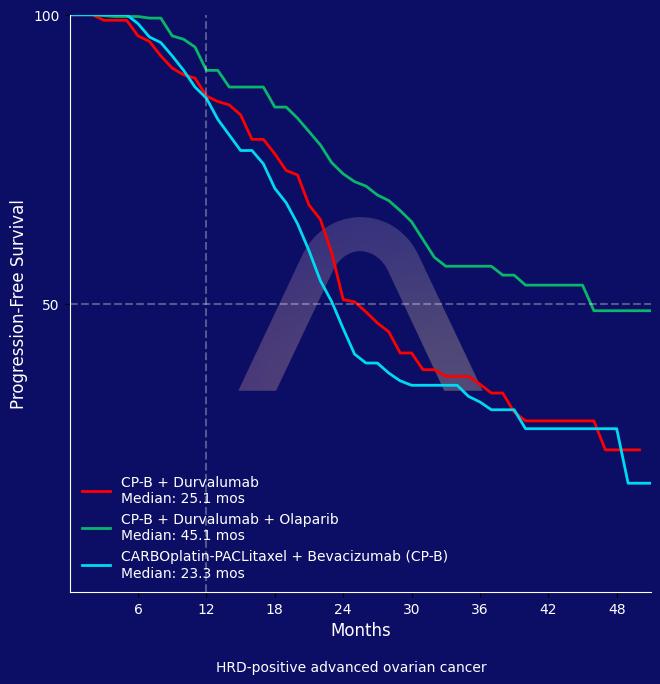
This phase III DUO-O study in newly diagnosed advanced-stage ovarian cancer patients without tumor BRCA mutation showed that adding durvalumab plus olaparib to carboplatin/paclitaxel and bevacizumab followed by maintenance durvalumab, bevacizumab, and olaparib significantly improved progression-free survival compared to carboplatin/paclitaxel plus bevacizumab followed by bevacizumab alone. The median progression-free survival in the durvalumab plus olaparib arm reached 45.1 months in the homologous recombination deficiency-positive population, representing the longest reported to date in this setting. No significant overall survival benefit was observed at interim analysis, and safety profiles were consistent with each individual agent’s known effects.
Study
|
Randomized, double-blind, placebo-controlled phase III trial [DUO-O, NCT03737643] |
| Newly diagnosed advanced-stage ovarian cancer without tumor BRCA mutation |
| Carboplatin/paclitaxel plus bevacizumab (CP-B) followed by bevacizumab (control; n=378), CP-B plus durvalumab followed by bevacizumab plus durvalumab (durvalumab arm; n=374), or same plus olaparib maintenance (durvalumab olaparib arm; n=378)
|
Efficacy
|
PFS non-tBRCAm ITT: 25.1 mos vs. 20.6 mos vs. 19.3 mos (durvalumab-olaparib arm vs. durvalumab arm vs. control) (Only significant for durvalumab-olaparib arm: HR 0.61 [0.51-0.73]) |
| PFS non-tBRCAm HRD-positive: 45.1 mos vs 25.1 mos vs. 23.3 mos (Only significant for durvalumab-olaparib arm: HR 0.46 [0.33-0.65])
|
Safety
|
Grade >=3 AEs: neutropenia (31.0% vs 27.6% vs 25.5%), anemia (24.6% vs 7.8% vs 7.7%), leukopenia (8.2% vs 5.1% vs 4.3%), thrombocytopenia (6.1% vs 3.8% vs 4.0%), hypertension (7.4% vs 8.8% vs 11.2%) |
| Grade 5 AEs: 2.1% vs 2.4% vs 1.1% (one AML in durvalumab + olaparib arm attributable to olaparib) |
| New primary malignancies: 1.1% vs 0.3% vs 0.8%
|
Ann Oncol. Published online 9 December 2025
http://doi.org/10.1016/j.annonc.2025.11.020
Reviewed by Ulas D. Bayraktar, MD on Apr 9, 2026