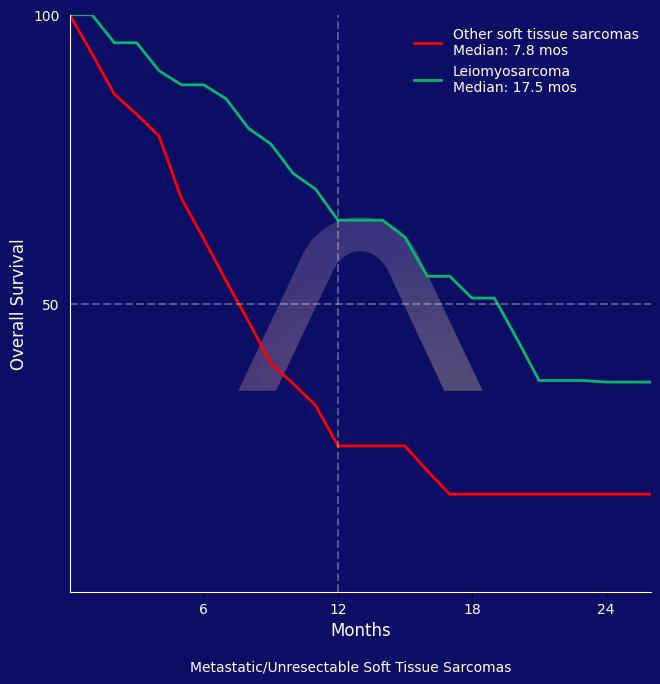
Cabozantinib and temozolomide combination demonstrated promising clinical benefit in patients with unresectable or metastatic leiomyosarcoma. The treatment showed manageable adverse events without any new safety concerns, and the progression-free survival was favorable compared to existing therapies.
Study
|
Single-arm, lead-in phase 2 trial [NCT04200443] |
| Unresectable or metastatic leiomyosarcoma (Cohort 1) and other soft tissue sarcomas (Cohort 2) |
| Cabozantinib and temozolomide (n=72)
|
Efficacy
|
PFS at 12 weeks: 74% (Cohort 1, leiomyosarcoma) |
| Median PFS Cohort 1: 6.3 mos |
| Median PFS Cohort 2: 3.0 mos |
| Median OS Cohort 1: 19.2 mos |
| Median OS Cohort 2: 7.7 mos
|
Safety
|
Grade >=3 AE: platelet count decrease (30%), neutrophil count decrease (18%), hypertension (10%), diarrhoea (8%) |
| No treatment-related deaths
|
Lancet Oncol. Published online January 16, 2026
http://doi.org/10.1016/S1470-2045(25)00654-0
Reviewed by Ulas D. Bayraktar, MD on Jan 21, 2026