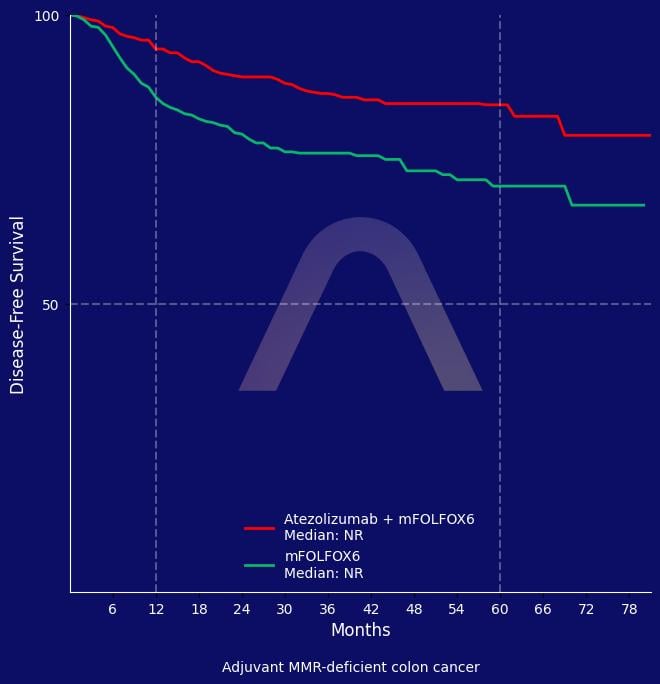
In this phase 3 randomized trial, the addition of atezolizumab to mFOLFOX6 significantly improved 3-year disease-free survival compared to mFOLFOX6 alone in patients with resected stage III mismatch repair–deficient colon cancer. No significant difference in overall survival was observed at median 45.8-month follow-up. The combination had a higher rate of grade 3 or 4 adverse events but was consistent with known safety profiles.
Study
|
Phase 3, randomized trial [ATOMIC; NCT02912559] |
| Resected stage III mismatch repair–deficient (dMMR) colon cancer |
| Atezolizumab plus modified FOLFOX6 (mFOLFOX6) (n=355) vs mFOLFOX6 alone (n=357) for 6 months, with atezolizumab continued as monotherapy to 12 months total
|
Efficacy
|
DFS @ 3yrs: 86.3% vs 76.2% (Atezo + mFOLFOX6 vs. FOLFOX6 alone)(HR 0.50 [0.35-0.73]) |
| OS @ 5yrs: 89.7% vs 87.9% (HR 0.90 [0.55-1.47])
|
Safety
|
Grade 3 or 4 adverse events: 84.1% vs 71.9% (atezolizumab+mFOLFOX6 vs mFOLFOX6) |
| Grade 3 or 4 AEs: Hematologic (46.8% vs 38.6%), fatigue (10.1% vs 3.3%)
|
N Engl J Med 2026;394:1155-66
http://doi.org/10.1056/NEJMoa2507874
Reviewed by Ulas D. Bayraktar, MD on Apr 9, 2026