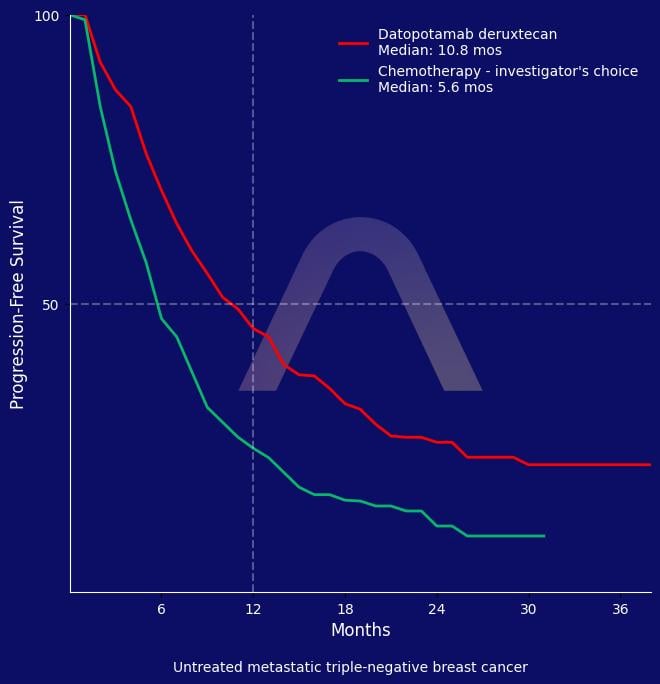
This phase III trial demonstrated that datopotamab deruxtecan significantly improved progression-free and overall survival compared with chemotherapy in patients with previously untreated metastatic triple-negative breast cancer who were ineligible for immunotherapy. The drug showed higher objective and complete response rates and longer duration of response. Rates of serious and grade 3 or higher treatment-related adverse events were similar to chemotherapy, with fewer discontinuations due to adverse events.
Study
|
Randomised, open-label, international, phase III trial [TROPION-Breast02] |
| Previously untreated, locally recurrent inoperable or metastatic triple-negative breast cancer in patients ineligible for immunotherapy |
| Datopotamab deruxtecan (n=323) vs investigator’s choice of chemotherapy (n=321)
|
Efficacy
|
ORR: 63% vs 29% (datopotamab vs ICC) (OR 4.24 [3.03-5.95]) |
| CR: 9% vs 2% |
| Median DoR: 12.3 mos vs 7.1 mos |
| Median PFS: 10.8 mos vs 5.6 mos (HR 0.57 [0.44-0.73]) |
| Median OS: 23.7 mos vs 18.7 mos (HR 0.79 [0.64-0.98])
|
Safety
|
Grade >=3 TRAEs: stomatitis (8% vs 9%), alopecia (NA vs 31%), fatigue (3% vs 3%), keratitis (2% vs NA), neutropenia (12% vs 29%), amylase increased (6% vs NA), diarrhea (0% vs 13%), peripheral neuropathy (0% vs 2%), pneumonia (1% vs 1%) |
| Serious TRAEs: 9% vs 8% |
| TRAEs leading to discontinuation: 4% vs 7%
|
Ann Oncol 2026. Published online 2026-03-28
http://doi.org/10.1016/j.annonc.2026.03.008
Reviewed by Ulas D. Bayraktar, MD on Apr 9, 2026