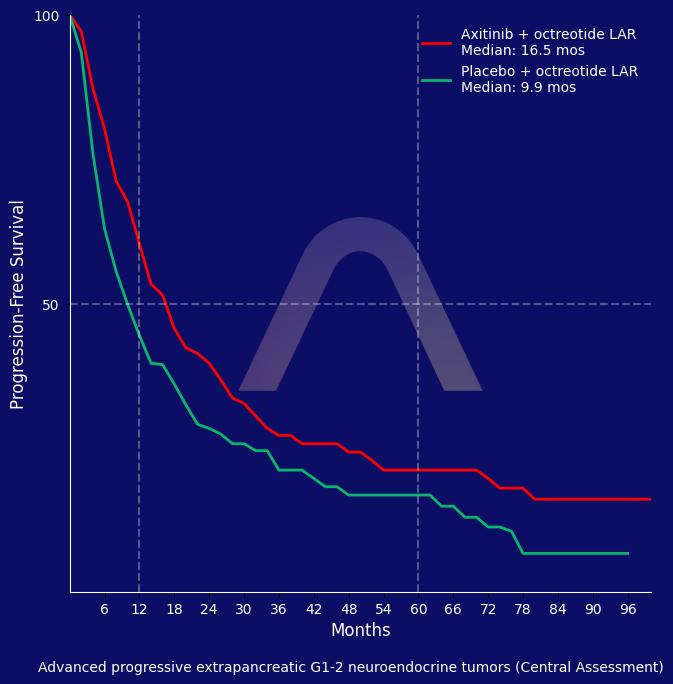
Axitinib combined with octreotide LAR in patients with advanced progressive extrapancreatic neuroendocrine tumors showed an increase in progression-free survival assessed by blinded central review but not by investigator assessment. The objective response rate was higher for axitinib compared to placebo. The treatment was associated with manageable toxicities.
Study
|
Randomized, double-blind, placebo-controlled, phase III study [AXINET, GETNE 1107] |
| Unresectable/metastatic progressive G1-2 extrapancreatic neuroendocrine tumors |
| Axitinib (n=126) vs placebo (n=130) with octreotide LAR until progression or unacceptable toxicity
|
Efficacy
|
ORR: 17.5% vs 4.6% (axitinib vs. placebo) |
| Investigator-assessed mPFS: 17.2 mos vs 13.1 mos (HR 0.86 [0.65-1.15]) |
| BICR-assessed mPFS: 16.6 mos vs 9.9 mos (HR 0.71 [0.54-0.94]) |
| Investigator-assessed ORR: 17.5% vs 4.6% |
| BICR-assessed ORR: 12.8% vs 3.2%
|
Safety
|
Grade >=3 AEs: hypertension (24% vs 9.2%), diarrhea (13.6% vs 1.5%) |
| Serious AEs: axitinib (38.4%) vs placebo (23.1%) |
| Treatment discontinuations due to TRAEs: 18.3% vs 3.1%
|
J Clin Oncol. Published online February 20, 2026
http://doi.org/10.1200/JCO-25-01808
Reviewed by Ulas D. Bayraktar, MD on Mar 11, 2026