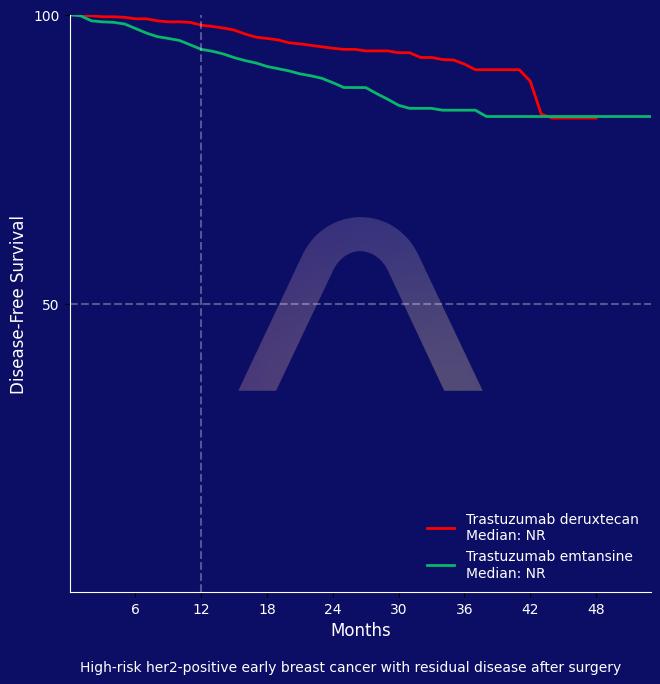
In this phase 3 trial, trastuzumab deruxtecan significantly improved invasive disease-free survival compared to trastuzumab emtansine in patients with residual HER2-positive early breast cancer. The safety profile showed higher rates of interstitial lung disease with T-DXd, which requires careful monitoring.
Study
|
Phase 3, open-label, international, randomized trial [DESTINY-Breast05] |
| Residual HER2-positive early breast cancer after neoadjuvant therapy |
| T-DXd (n=818) vs T-DM1 (n=817)
|
Efficacy
|
3-year invasive disease-free survival: 92.4% vs 83.7% (HR 0.47 [0.34-0.66]) |
| 3-year disease-free survival: 92.3% vs 83.5% (HR 0.47 [0.34-0.66])
|
Safety
|
Grade >=3 AEs: 50.6% vs 51.9% |
| Interstitial lung disease: 9.6% vs 1.6% |
| Discontinuation due to AEs: 17.9% vs 12.9% |
| Drug-related deaths: 0.4% vs 0.6%
|
N Engl J Med 2026;394:845-57
Loibl S, Park YH, Shao Z Trastuzumab Deruxtecan in Residual HER2-Positive Early Breast Cancer
http://doi.org/10.1056/NEJMoa2514661
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026