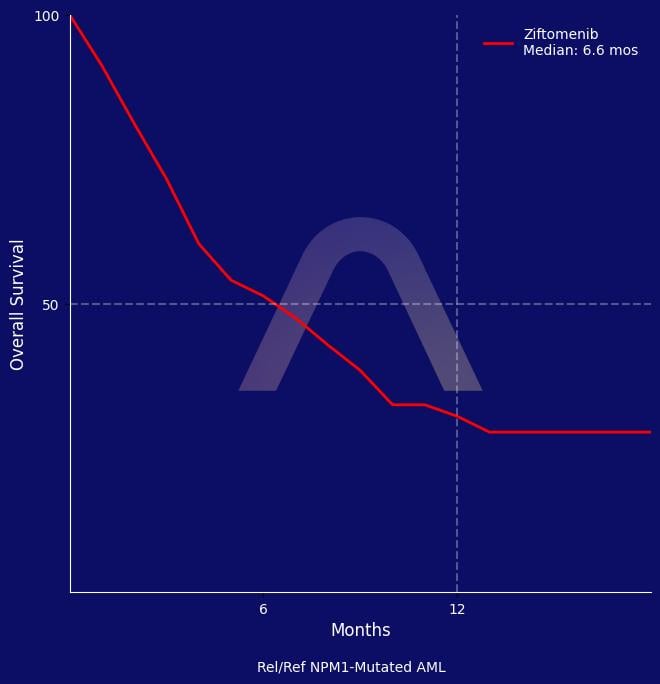
Ziftomenib monotherapy led to a complete remission rate of 22% with a median overall survival of 6.6 months among relapsed/refractory NPM1-mutated AML patients. It was well tolerated with manageable differentiation syndrome and low rates of severe adverse events.
Study
|
Phase II study [KOMET-001] |
| Relapsed/refractory NPM1-mutated AML patients |
| Ziftomenib 600 mg daily (n=92)
|
Efficacy
|
CR/CRh: 22% [14-32] |
| mOS: 6.6 mos [3.6-8.6] |
| ORR: 33% [23-43]
|
Safety
|
Grade >=3 AEs: Febrile neutropenia (26%), anemia (20%), thrombocytopenia (20%) |
| Differentiation syndrome (25%) |
| Discontinuations due to AEs: 3%
|
J Clin Oncol 2025;43:3381-3390
http://doi.org/10.1200/JCO-25-01694
Reviewed by Ulas D. Bayraktar, MD on Nov 16, 2025