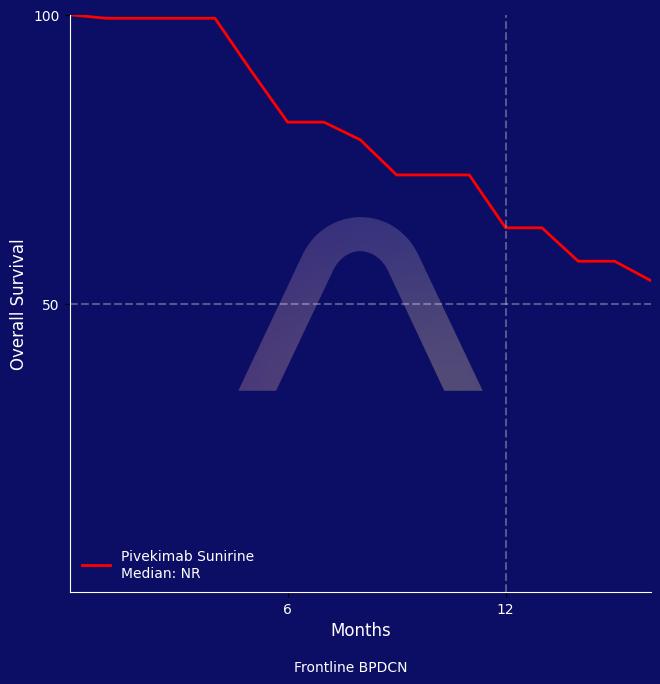
Pivekimab Sunirine showed high response rates in patients with frontline de novo blastic plasmacytoid dendritic cell neoplasm, with a composite complete response rate of 75% and a median overall survival of 16.6 months. In relapsed or refractory cases, the response rate was lower at 14% with a median overall survival of 5.8 months. The treatment was generally well tolerated with manageable safety concerns, making it a promising option for patients with limited treatment alternatives.
Study
|
Phase I/II, open-label, multicenter study [NCT03386513] |
| Adult patients with frontline or relapsed/refractory BPDCN |
| Pivekimab Sunirine 0.045 mg/kg once every 3 weeks
|
Efficacy
|
CR + clinical CR: 75% in frontline de novo vs 14% in relapsed/refractory |
| mOS: 16.6 mos vs 5.8 mos
|
Safety
|
Serious AEs: Pneumonia (6%), febrile neutropenia (5%) |
| VOD: 2 cases reported
|
J Clin Oncol. Published online February 11, 2026
http://doi.org/10.1200/JCO-25-02083
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026