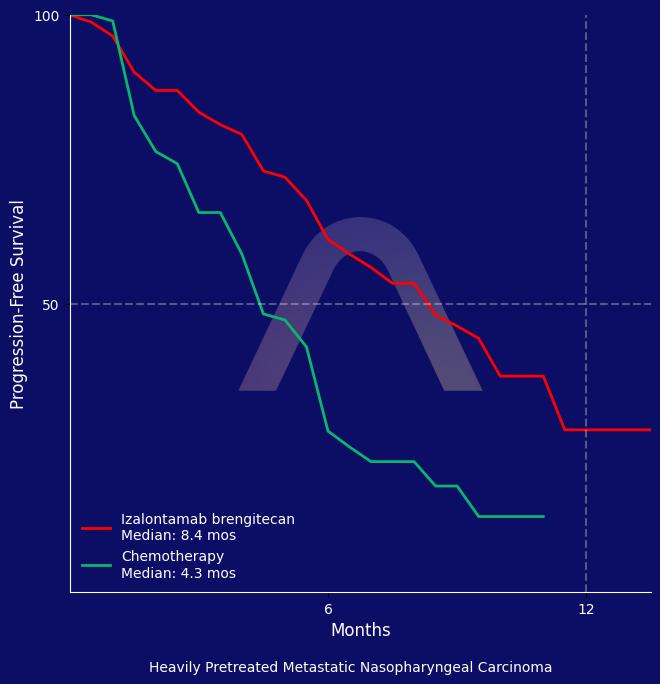
Izalontamab brengitecan significantly improved objective response rate and progression-free survival compared to chemotherapy in heavily pretreated nasopharyngeal carcinoma patients. The therapy’s safety profile was manageable, although haematological toxic effects were common. Further analysis is needed to evaluate the overall survival benefit.
Study
|
Multicentre, randomized, open-label, phase 3 study [NCT06118333] |
| Heavily pretreated recurrent or metastatic nasopharyngeal carcinoma |
| Izalontamab brengitecan (n=191) vs chemotherapy (n=195)
|
Efficacy
|
ORR: 54.6% vs 27.0% |
| mPFS: 8.38 mos vs 4.34 mos (HR 0.44 [0.32-0.62]) |
| Overall survival data were immature at the time of data cutoff.
|
Safety
|
Grade >=3 AEs: anaemia (50% vs 10%), decreased WBC count (43% vs 44%), decreased platelet count (43% vs 7%), decreased neutrophil count (38% vs 41%) |
| Serious AEs: 43% vs 27% |
| Treatment-related deaths: 2% vs None reported
|
Lancet. Published online October 19, 2025
Yang Y, Zhou H, Tang L New Drug: Izalontamab Brengitecan for Metastatic Nasopharyngeal Carcinoma
http://doi.org/10.1016/S0140-6736(25)01954-3
Reviewed by Ulas D. Bayraktar, MD on Nov 15, 2025