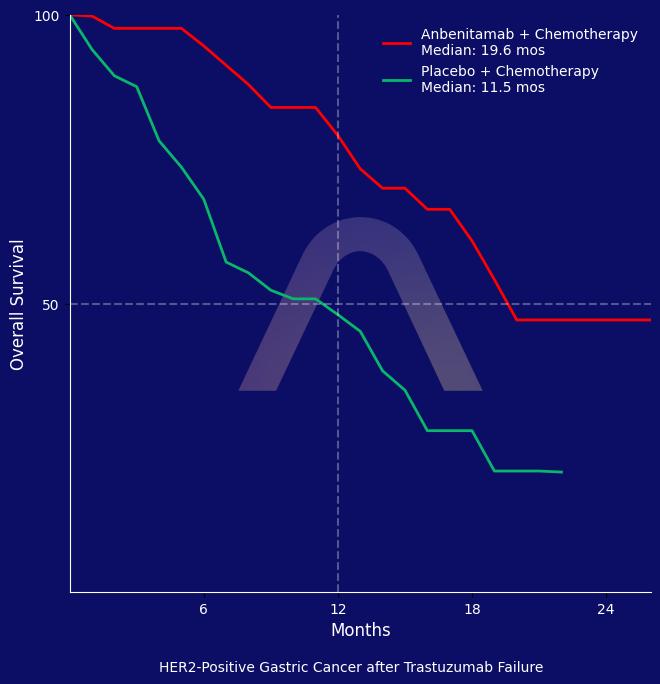
The study illustrates that anbenitamab plus chemotherapy significantly improves progression-free survival and overall survival in patients with HER2-positive gastric cancer who have progressed after trastuzumab therapy. The treatment was well tolerated, with a higher overall response rate observed compared to chemotherapy alone.
Study
|
Randomized, double-blind, phase III trial [NCT05427383] |
| HER2-positive gastric or gastroesophageal junction adenocarcinoma after trastuzumab + chemotherapy failure |
| Anbenitamab+Chemotherapy (n=95) vs Placebo+Chemotherapy (n=93) every 3 weeks
|
Efficacy
|
ORR: 56% vs 11% (anbenitamab vs. placebo) |
| mPFS: 7.1 mos vs 2.7 mos (HR 0.25 [0.17-0.39]) |
| mOS: 19.6 mos vs 11.5 mos (HR 0.29 [0.17-0.50])
|
Safety
|
Grade >=3 AE: neutropenia (30% vs 22%), leukopenia (21% vs 25%), anemia (18% vs 11%) |
| Serious AE: 26% vs 23% |
| Treatment discontinuation: 11% vs 2% |
| Treatment-related deaths: 0 vs 5%
|
Ann Oncol. Published online Feb 2026
http://doi.org/10.1016/j.annonc.2026.01.006
Reviewed by Ulas D. Bayraktar, MD on Feb 15, 2026