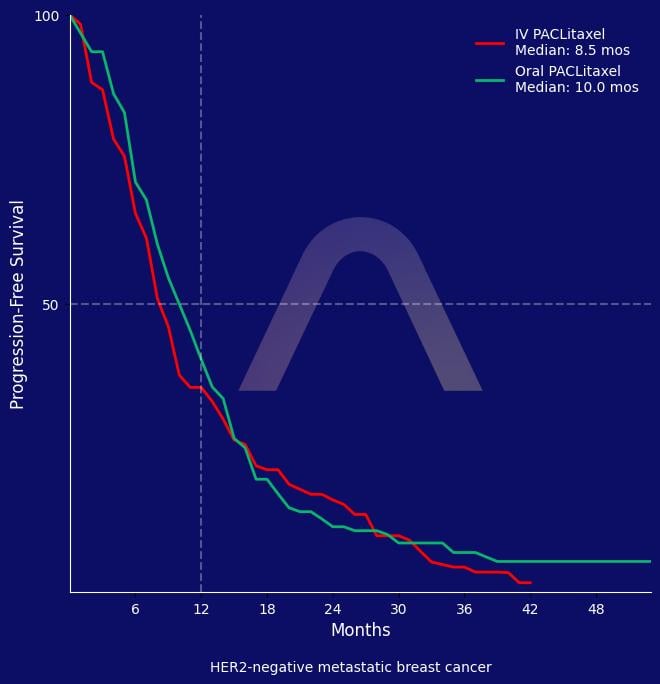
In this multinational phase III trial, oral paclitaxel DHP107 demonstrated non-inferior progression-free survival compared to intravenous weekly paclitaxel in patients with HER2-negative recurrent or metastatic breast cancer. Overall survival and tumor response rates were comparable, with a manageable safety profile noted for DHP107 including higher neutropenia and gastrointestinal toxicities but less peripheral neuropathy and hypersensitivity. The oral formulation offers a convenient alternative to intravenous treatment.
Study
|
Multinational, multicenter, open-label, randomized phase III non-inferiority trial [OPTIMAL, NCT03315364] |
| HER2-negative recurrent or metastatic breast cancer, chemotherapy-naive in metastatic setting |
| DHP107 (200 mg/m2 orally twice daily on days 1, 8, 15 of 28-day cycle) (n=277) vs intravenous paclitaxel (80 mg/m2 days 1, 8, 15 of 28-day cycle) (n=272)
|
Efficacy
|
ORR: 43.3% vs 38.8% |
| Median PFS: 10.0 mos (95% CI 9.1-11.2) vs 8.5 mos (95% CI 7.4-9.3) (HR 0.869 [0.707-1.068]) |
| Median OS: 32.6 mos (95% CI 26.0-34.9) vs 31.8 mos (95% CI 24.4-36.3) (HR 0.967 [0.762-1.227])
|
Safety
|
Grade >=3 AEs: Neutropenia (67.1% vs 29.7%), febrile neutropenia (6.1% vs 0.8%), nausea (1.1% vs 0.4%), diarrhea (0.7% vs 1.5%), vomiting (0.7% vs 0.4%), peripheral neuropathy (1.8% vs 0.4%), hypersensitivity (0% vs 0.4%), injection site reaction (0.4% vs 0.4%) |
| Grade >=3 adverse events more frequent with DHP107; SAEs 17.7% vs 9.9%; discontinuations due to AEs 12.3% vs 8.7% |
| Treatment-related deaths: 0% vs 0.4%
|
Ann Oncol. Published online 2026-03
http://doi.org/10.1016/j.annonc.2026.03.002
Reviewed by Ulas D. Bayraktar, MD on Apr 9, 2026