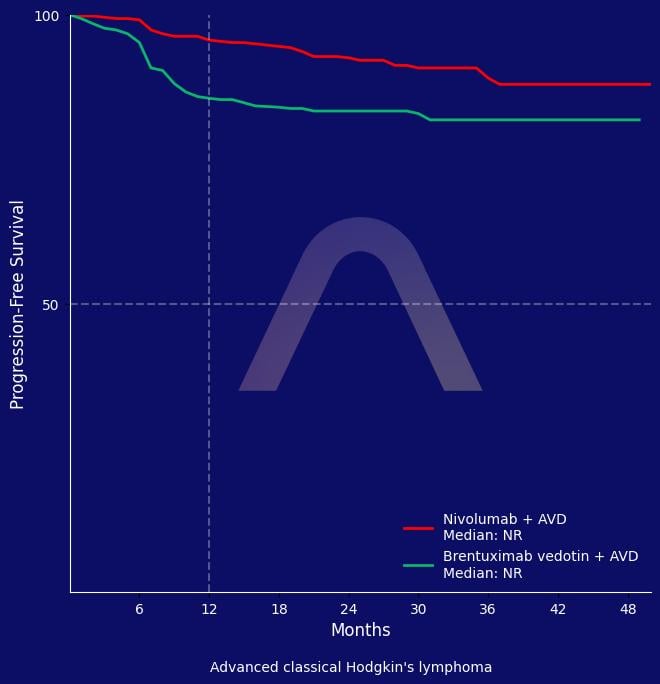
In this phase 3 trial of patients with advanced-stage classic Hodgkin’s lymphoma, nivolumab combined with AVD chemotherapy resulted in significantly improved progression-free survival compared to brentuximab vedotin combined with AVD. Nivolumab plus AVD also demonstrated a better side-effect profile, with fewer treatment discontinuations, less peripheral neuropathy, and fewer immune-related adverse events. The low use of radiotherapy and the favorable safety and efficacy support nivolumab plus AVD as a preferred primary treatment option for these patients.
Study
|
Phase 3, multicenter, open-label, randomized trial [S1826] |
| Stage III or IV newly diagnosed Hodgkin’s lymphoma |
| Nivolumab+AVD (n=487) vs Brentuximab vedotin+AVD (n=483)
|
Efficacy
|
2-year PFS: 92% vs 83% (N-AVD vs. B-AVD) (HR 0.45 [0.30-0.65]) |
| 2-year EFS: 90% vs 81% (HR 0.50 [0.36-0.71]) |
| 2-year OS: 99% vs 98% (HR 0.39 [0.15-1.03])
|
Safety
|
Any grade AEs: Neutropenia (48% vs 26%), Peripheral sensory neuropathy (9% vs 32%), Treatment discontinuation (7.6% vs 12%), Hypothyroidism (7% vs <1%), Hyperthyroidism (3% vs 0%)
|
N Engl J Med 2024;391:1379-89
http://doi.org/10.1056/NEJMoa2405888
Reviewed by Ulas D. Bayraktar, MD on Apr 9, 2026