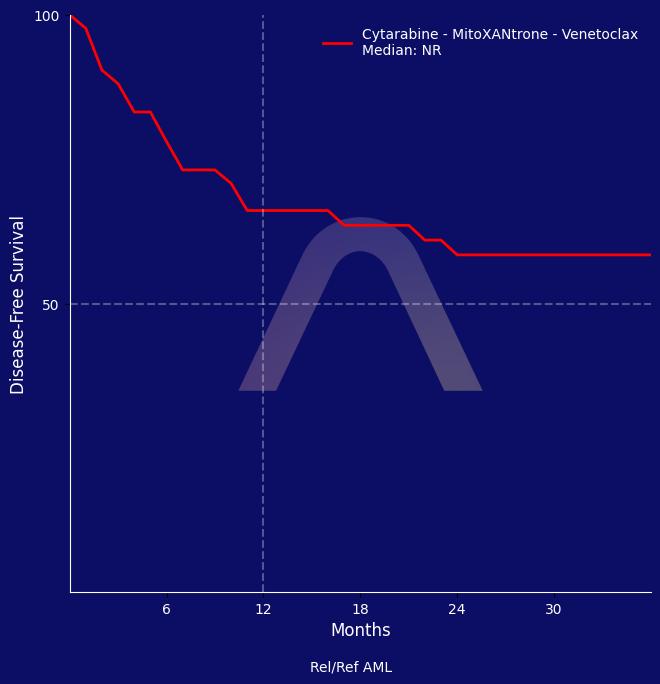
The RELAX trial demonstrated that the combination of venetoclax with high-dose cytarabine and mitoxantrone is effective and safe for treating relapsed or refractory acute myeloid leukaemia. The treatment achieved a composite complete remission rate of 75%, and showed a favorable safety profile with potentially manageable adverse effects. This regimen may serve as a bridge to allogeneic transplantation for medically fit patients.
Study
|
Multicentre, open-label, single-arm, phase 1/2 trial [RELAX] |
| Relapsed or refractory acute myeloid leukaemia, aged 18-75 |
| Venetoclax 400 mg daily (days 1-14), mitoxantrone 10 mg/m² (days 5-7), cytarabine dose escalation
|
Efficacy
|
Composite complete remission: 75% |
| 12-month RFS: 66% |
| 24-month RFS: 58% |
| 12-month OS: 65% |
| 24-month OS: 56%
|
Safety
|
Grade >=3 AEs: febrile neutropenia (53%), pneumonia (13%), sepsis (13%) |
| Serious AEs: sepsis (n=6), pneumonia (n=2) |
| Potential treatment-related deaths: 4 (7%), including sepsis (3) and pneumonia (1)
|
Lancet Haematol 2026;13:e157-68
http://doi.org/10.1016/S2352-3026(25)00358-8
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026