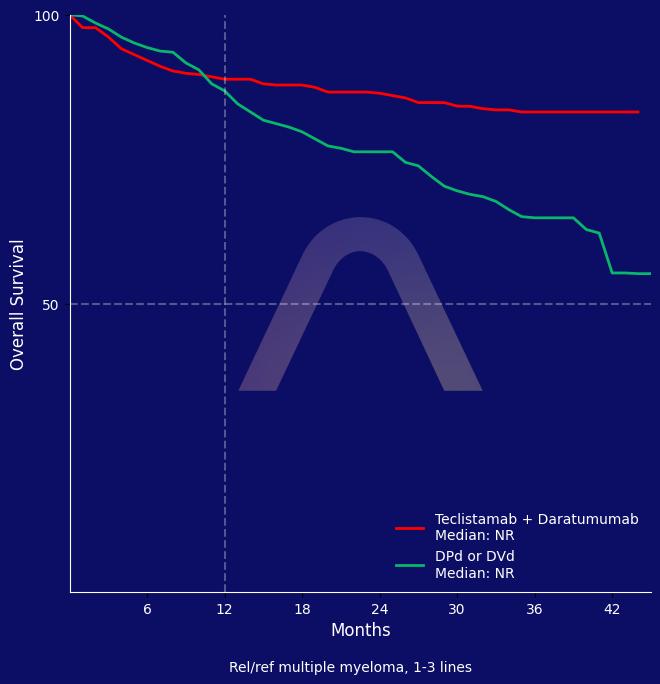
The study demonstrated significantly longer progression-free survival with teclistamab-daratumumab compared to DPd or DVd, with a hazard ratio of 0.17. The combination also achieved higher overall response rates and complete responses. A higher rate of infection-related deaths was observed in the teclistamab group.
Study
|
Phase 3, multicenter, open-label trial [NCT05083169] |
| Relapsed or refractory multiple myeloma, 1-3 previous lines of therapy |
| Teclistamab-Daratumumab (n=291) vs DPd or DVd (n=296)
|
Efficacy
|
ORR: 89.0% vs 75.3% (Teclistamab-daratumumab vs. DPd/DVd) |
| CR or better: 81.8% vs 32.1% |
| MRD negativity: 58.4% vs 17.1% |
| 3yr PFS: 83.4% vs 29.7% (HR 0.17 [0.12-0.23]) |
| 3yr OS:83.3% vs. 65%
|
Safety
|
Grade >=3 AEs: Neutropenia (75.6% vs 78.6%), Anemia (20.5% vs 17.2%), Thrombocytopenia (19.4% vs 23.4%) |
| All cytokine release syndromes were Grade 1-2 |
| Serious AEs: 70.7% vs 62.4% |
| Deaths from AEs: 7.1% vs 5.9%
|
N Engl J Med 2026;394:739-52
http://doi.org/10.1056/NEJMoa2514663
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026