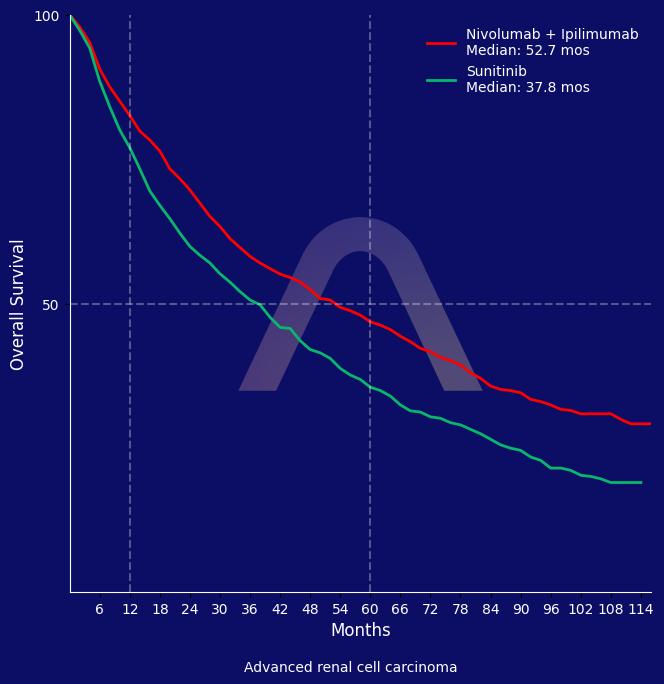
Nivolumab plus ipilimumab demonstrated a significant survival benefit and higher response rates compared to sunitinib for first-line treatment of advanced renal cell carcinoma, with a median survival of 52.7 months. The combination also had fewer grade 3 or higher adverse events but a higher discontinuation rate due to adverse events.
Study
|
Phase III, open-label, randomized trial [CheckMate 214, NCT02231749] |
| Previously untreated advanced or metastatic renal cell carcinoma with a clear cell component |
| Nivolumab+Ipilimumab (n=547) vs Sunitinib (n=535) until progression or unacceptable toxicity
|
Efficacy
|
ORR: 39.5% vs 33.0%(nivo + ipi vs. sunitinib) |
| CR: 12.0% vs 3.5% |
| mPFS: 12.4 mos vs 12.3 mos (HR 0.88 [0.76-1.04]) |
| mOS: 52.7 mos vs 37.8 mos (HR 0.71 [0.62-0.82]) |
| 9-yr OS: 31.4% vs. 19.5% |
| mOS in interm/poor-risk pts: 46.7 mos vs. 26.0 mos (HR 0.69 [0.59-0.81])
|
Safety
|
Grade >=3 AEs: 48.6% vs 64.1% |
| Discontinuation due to AEs: 23.8% vs 13.3% |
| Deaths due to study drug: 1.5% vs 0.9%
|
Ann Oncol. Published online 2026
http://doi.org/10.1016/j.annonc.2026.02.017
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026