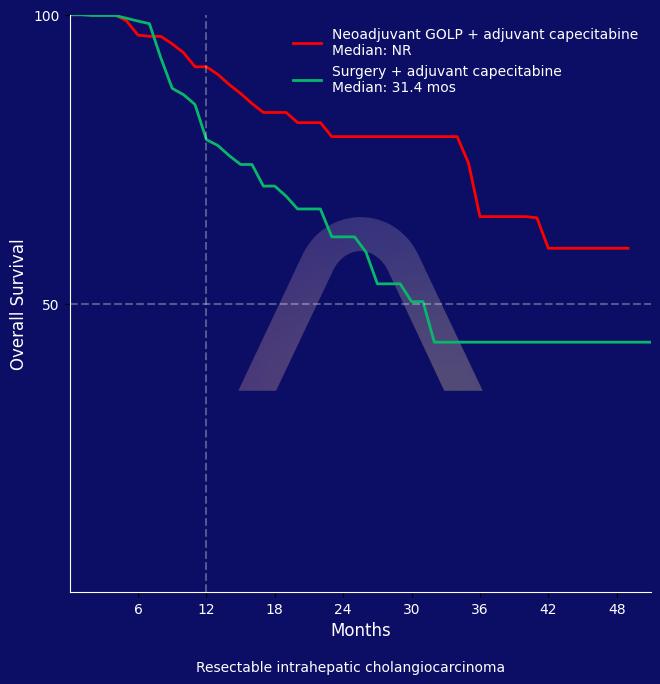
The study demonstrated that neoadjuvant GOLP significantly extended event-free survival compared to the control group in patients with resectable high-risk intrahepatic cholangiocarcinoma. The overall survival difference did not meet the pre-specified criterion of p = 0.0019. The survival advantage was accompanied by a manageable safety profile with few high-grade adverse events, emphasizing the potential of the GOLP regimen in this patient population.
Study
|
Phase 2-3 randomized, controlled, open-label, multicenter trial [ZSAB-neoGOLP] |
| Patients with resectable high-risk intrahepatic cholangiocarcinoma |
| Neoadjuvant GOLP (n=88) vs surgery alone (n=90); both followed by adjuvant capecitabine
|
Efficacy
|
PR: 55% in neoadjuvant group |
| R0 resection: 95% vs. 93% (neoadjuvant GOLP vs. no neoadjuvant therapy) |
| 24-mo OS: 79% vs 61% (HR 0.43 [0.23-0.79]) |
| Median EFS: 18.0 mos vs 8.7 mos
|
Safety
|
Grade >=3 AEs during neoadjuvant: neutropenia (17%) |
| During adjuvant: bilirubin increased (3% vs 1%)
|
N Engl J Med 2026;394:983-95
Shi GM, Huang XY, Liang F Neoadjuvant GOLP in Resectable High-Risk Intrahepatic Cholangiocarcinoma
http://doi.org/10.1056/NEJMoa2513918
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026