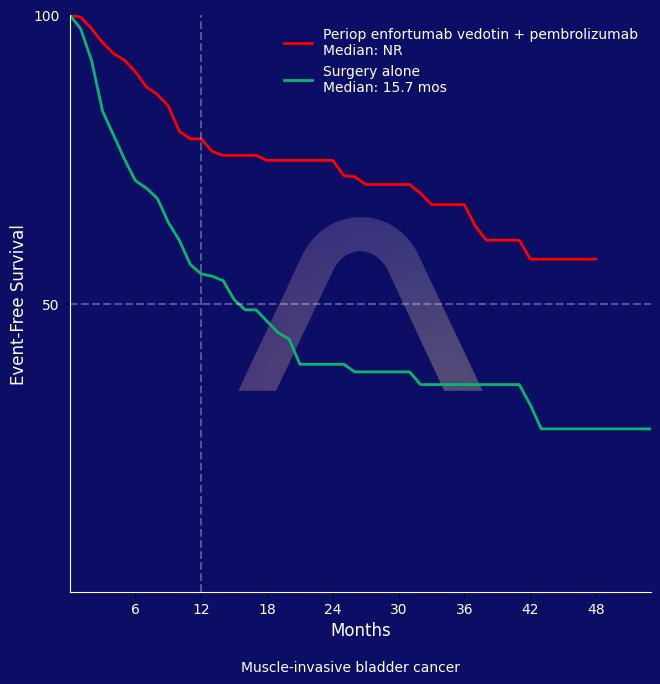
Perioperative enfortumab vedotin and pembrolizumab significantly improved event-free and overall survival compared to surgery alone in muscle-invasive bladder cancer patients ineligible for cisplatin. The treatment also led to a higher rate of pathological complete response. Adverse events were more frequent with the drug combination, though the safety profile was consistent with expectations.
Study
|
Phase 3, open-label, randomized trial [KEYNOTE-905/EV-303] |
| Muscle-invasive bladder cancer ineligible for cisplatin |
| Enfortumab vedotin+pembrolizumab (n=170) vs surgery alone (n=174).
|
Efficacy
|
pCR: 57.1% vs 8.6% (periop therapy vs. surgery alone) |
| 2-yr EFS: 74.7% vs 39.4% (HR 0.40 [0.28-0.57]) |
| 2-yr OS: 79.7% vs 63.1% (HR 0.50 [0.33-0.74])
|
Safety
|
Grade >=3 AEs: 71.3% vs 45.9%. |
| Serious AEs: 58.1% vs 40.9%. |
| Deaths related to AEs: 7.8% vs 5.7%.
|
N Engl J Med. Published online February 18, 2026
http://doi.org/10.1056/NEJMoa2511674
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026