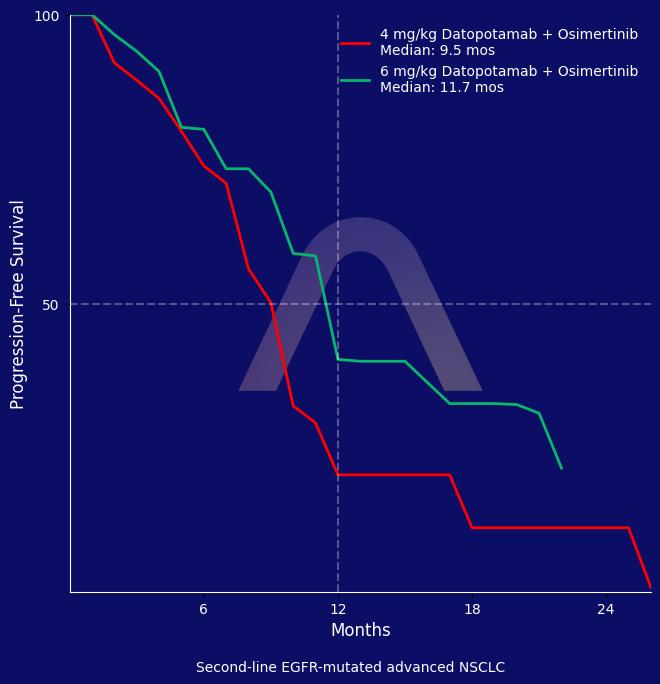
The combination of osimertinib and datopotamab deruxtecan showed clinical benefit in patients with EGFR-mutated advanced NSCLC who progressed on first-line osimertinib. The 6 mg/kg dose demonstrated higher efficacy but had more frequent adverse events, which could be managed with careful monitoring and dose adjustments. The study supports further investigation of the combination at the 6 mg/kg dose.
Study
|
Phase II open-label study [ORCHARD] |
| EGFR-mutated advanced NSCLC post-progression on first-line osimertinib |
| Osimertinib 80 mg QD plus Datopotamab Deruxtecan 4 mg/kg (n=35) vs 6 mg/kg (n=34)
|
Efficacy
|
ORR: 43% vs 36% (4 mg/kg vs. 6 mg/kg) |
| mPFS: 9.5 mos (7.2-9.8) vs 11.7 mos (8.3-21.7) |
| mOS: 19.8 mos (13.5-23.3) vs 26.2 mos (14.8-NC)
|
Safety
|
Grade >=3 AEs: 49% vs 76% |
| ILd/pneumonitis: 3% vs 15% |
| Dose reductions: 23% vs 59%
|
Ann Oncol. Published online 2026
http://doi.org/10.1016/j.annonc.2026.02.014
Reviewed by Ulas D. Bayraktar, MD on Mar 12, 2026