Study
| Randomised, double-blind, phase 3 trial [POD1UM-303/InterAACT-2]. |
| Patients with inoperable locally recurrent or metastatic squamous cell carcinoma of the anal canal, aged >=18, ECOG PS 0 or 1. |
| Retifanlimab+carboplatin+paclitaxel (n=154) vs placebo+carboplatin+paclitaxel (n=154) for up to 1 year. |
Efficacy
| ORR: 56% vs 44% (p=0.013) |
| Median PFS: 9.3 mos vs 7.4 mos (HR 0.63 [0.47-0.84]) |
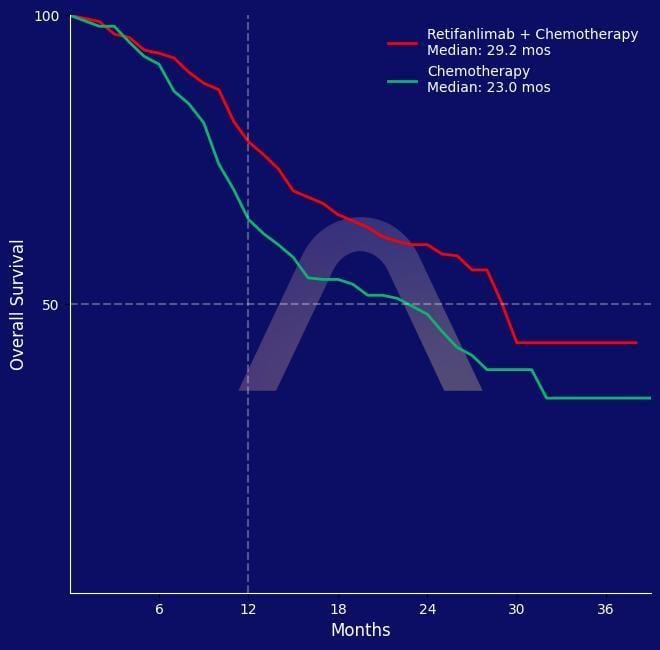
| Median OS: 29.2 mos vs 23.0 mos (HR 0.70 [0.49-1.01], p=0.027) |
Safety
| Grade >=3 AEs: 83% vs 75% |
| Serious AEs: 47% vs 39% |
| Neutropenia: 35% vs 30% |
| Anaemia: 20% vs 20% |
| Treatment-related deaths: 4 vs 1 |
Lancet 2025;405:2144-2152
http://doi.org/10.1016/S0140-6736(25)00631-2
Reviewed by Elvin Chalabiyev, MD on Sep 3, 2025